3.6 Valence Bond Theory
Valence Bond Theory (VBT), proposed by Heitler and London in 1927 and later developed by Pauling, provides a quantum mechanical explanation for the formation of covalent bonds. It successfully explains key molecular properties like bond energies, bond lengths, and the shapes of covalent molecules.
The core idea of VBT is that covalent bonds are formed by the overlapping of partially filled atomic orbitals from two different atoms. The two overlapping orbitals share a common region in space, but otherwise retain their individual identities.
Main Points of VBT
- A covalent bond is formed by the overlap of two half-filled atomic orbitals, one from each of the bonding atoms.
- The electrons in the overlapping orbitals must have opposite spins.
- The number of covalent bonds an atom can form is typically equal to the number of unpaired electrons in its outermost (valence) shell.
- The first bond formed by the direct overlap of two orbitals is a sigma () bond. Subsequent bonds (in double or triple bonds) are pi () bonds, formed by the overlap of additional orbitals.
- For a bond to form, the overlapping orbitals must have the same symmetry with respect to the bond axis (the imaginary line joining the nuclei of the two bonded atoms).
- The process of orbital overlap releases energy. The greater the extent of the overlap, the more energy is released, and the stronger the resulting bond.
Types of Overlapping and Nature of Covalent Bonds
The way atomic orbitals overlap determines the type of covalent bond formed. The two main types are sigma () and pi () bonds.
Sigma () Bond
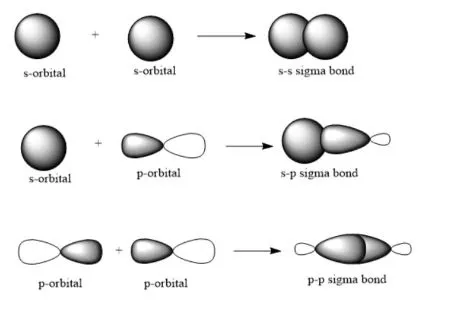
A sigma bond is formed by the direct, head-on overlap of atomic orbitals along the internuclear axis. The electron density in a sigma bond is concentrated directly between the two nuclei. All single bonds are sigma bonds.
Types of Overlap:
- overlap (e.g., in )
- overlap (e.g., in )
- head-on overlap (e.g., in )
Pi () Bond
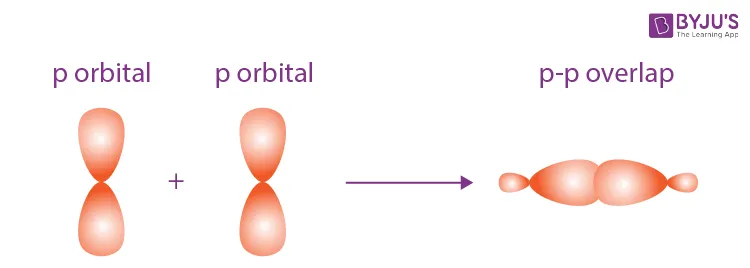
A pi bond is formed by the sideways or parallel overlap of atomic orbitals (typically p-orbitals) above and below the internuclear axis. The electron density in a pi bond is located in two regions, one above and one below the plane of the sigma bond.
Types of Overlap:
- parallel overlap
- parallel overlap
Multiple Bonds
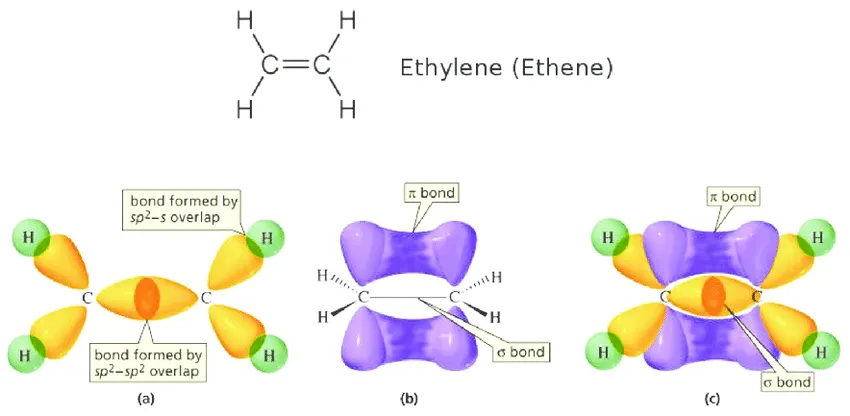
- A double bond consists of one sigma () bond and one pi () bond (e.g., Ethene, ).
- A triple bond consists of one sigma () bond and two pi () bonds (e.g., Acetylene, ).
Strength of Sigma and Pi Bonds
A sigma () bond is stronger than a pi () bond. This is because the head-on overlap in a sigma bond is more effective and results in a higher electron density concentrated between the nuclei, leading to a stronger attraction. The sideways overlap of a pi bond is less effective, and its electron density is more diffuse (spread out), making it a weaker and more easily broken bond.
Single Bond Formation
a) Formation of the Molecule
The electron configuration of a hydrogen atom is . A covalent bond forms when the half-filled orbital of one hydrogen atom overlaps head-on with the half-filled orbital of another hydrogen atom. This overlap creates a sigma () bond.
b) Formation of the Molecule
The electron configuration of fluorine is . It has one half-filled orbital, the orbital. The head-on overlap of the orbital from each fluorine atom forms a sigma () bond.
Figure 3.6.6: Orbital diagram for the formation of the molecule.
c) Formation of the Molecule
In the hydrogen fluoride molecule, the half-filled orbital of the hydrogen atom overlaps with the half-filled orbital of the fluorine atom. This head-on overlap results in a sigma () bond.
Multiple Bond Formation
a) Formation of the Molecule (Double Bond)
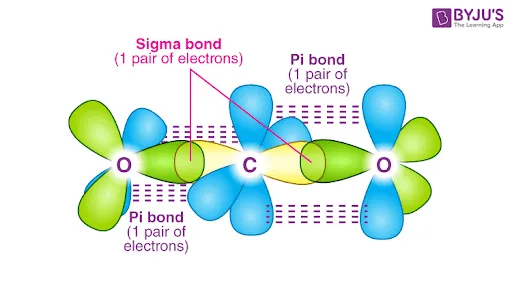
An oxygen atom () has two half-filled p-orbitals ().
- One pair of p-orbitals (e.g., ) overlaps head-on to form a sigma () bond.
- The other pair of p-orbitals () overlaps sideways (parallel) to form a pi () bond.
The combination results in a double bond () between the two oxygen atoms.
b) Formation of the Molecule (Triple Bond)
A nitrogen atom () has three half-filled p-orbitals ().
- The orbitals from each nitrogen atom overlap head-on to form a sigma () bond.
- The orbitals from each atom overlap sideways to form the first pi () bond.
- The orbitals from each atom also overlap sideways to form the second pi () bond.
This results in a triple bond () between the two nitrogen atoms.
| Bond Type | Composition |
|---|---|
| Single Bond | 1 bond |
| Double Bond | 1 + 1 bond |
| Triple Bond | 1 + 2 bonds |
For related concepts, see Shapes of Orbitals→, Rules of Electronic Configuration→, and Molecular Orbital Theory→.