3.5 Shapes of Molecules
Valence Shell Electron Pair Repulsion (VSEPR) Theory
The Valence Shell Electron Pair Repulsion (VSEPR) theory is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms.
The core principle is that electron pairs, being negatively charged, repel each other. They arrange themselves around the central atom in a way that maximizes the distance between them, thus minimizing the repulsive forces.
The final shape of a molecule is determined by the number of bonding pairs (electrons shared in a covalent bond) and lone pairs (non-bonding electrons) around the central atom.
Hierarchy of Electron Pair Repulsion
Lone pairs have a more concentrated electron charge cloud than bonding pairs, leading to stronger repulsion. The order of repulsion strength is:
- Lone Pair – Lone Pair (LP–LP) > Lone Pair – Bonding Pair (LP–BP) > Bonding Pair – Bonding Pair (BP–BP)
This hierarchy explains why molecules with lone pairs often have distorted bond angles compared to idealized geometries.
Note on Multiple Bonds: Double and triple bonds are treated as a single electron domain (super pair) in VSEPR theory for the purpose of determining geometry, though they occupy slightly more space than single bonds.
3.5.1 Shapes of Molecules with Two Electron Pairs
Molecules where the central atom is surrounded by two bonding pairs and no lone pairs () adopt a linear shape with a bond angle of .
Example: Beryllium Fluoride ()
- The central beryllium atom contributes two valence electrons.
- Each fluorine atom contributes one electron to the bond.
- The Lewis structure shows two single bonds from Be to F.
- To minimize repulsion, the two electron pairs are positioned on opposite sides of the central atom.
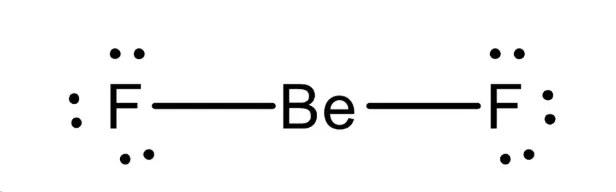
Example: Carbon Dioxide ()
- The central carbon atom forms two double bonds with oxygen atoms.
- Each double bond is treated as one electron domain → two electron domains total, no lone pairs.
- Result: linear, bond angle = .
Why is linear while is bent?
Both molecules have 3 atoms (same atomicity), but their shapes differ because of lone pairs:
| Molecule | Type | Bonding Pairs | Lone Pairs | Shape | Bond Angle |
|---|---|---|---|---|---|
| 2 | 0 | Linear | |||
| 2 | 2 | Bent |
The two lone pairs on oxygen in exert strong LP–LP and LP–BP repulsions, compressing the bond angle to .
3.5.2 Shapes of Molecules with Three Electron Pairs
Three Bonding Pairs ()
Molecules with three bonding pairs and no lone pairs have a trigonal planar geometry with bond angles of .
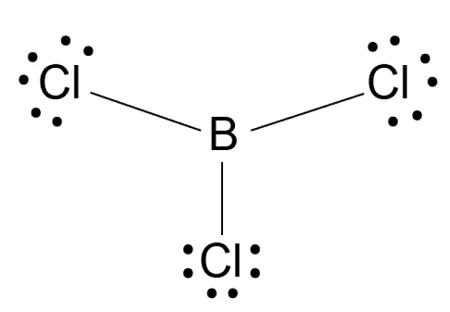
Example: Boron Trichloride ()
- The central boron atom contributes three valence electrons.
- The three bonding pairs repel each other equally, arranging themselves in a plane.
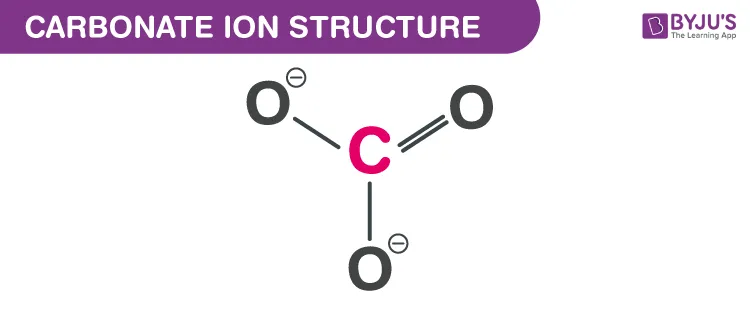
Example: Carbonate Ion ()
- The central carbon atom is bonded to three oxygen atoms. The overall structure is trigonal planar.
Two Bonding Pairs and One Lone Pair ()
Molecules with two bonding pairs and one lone pair have an electron geometry that is trigonal planar, but the molecular shape is bent or V-shaped. The bond angle is less than due to the stronger repulsion from the lone pair.
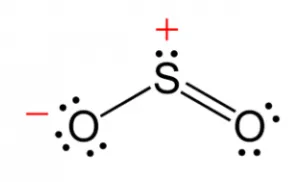
Example: Sulfur Dioxide ()
- The central sulfur atom has three electron domains: two double bonds and one lone pair.
- The lone pair–bonding pair (LP–BP) repulsion is greater than the bonding pair–bonding pair (BP–BP) repulsion, pushing the bonding pairs closer together.
3.5.3 Shapes of Molecules with Four Electron Pairs
Four Bonding Pairs ()
Molecules with four bonding pairs and no lone pairs have a tetrahedral geometry with bond angles of .
Example: Methane ()
- The central carbon atom is surrounded by four bonding electron pairs.
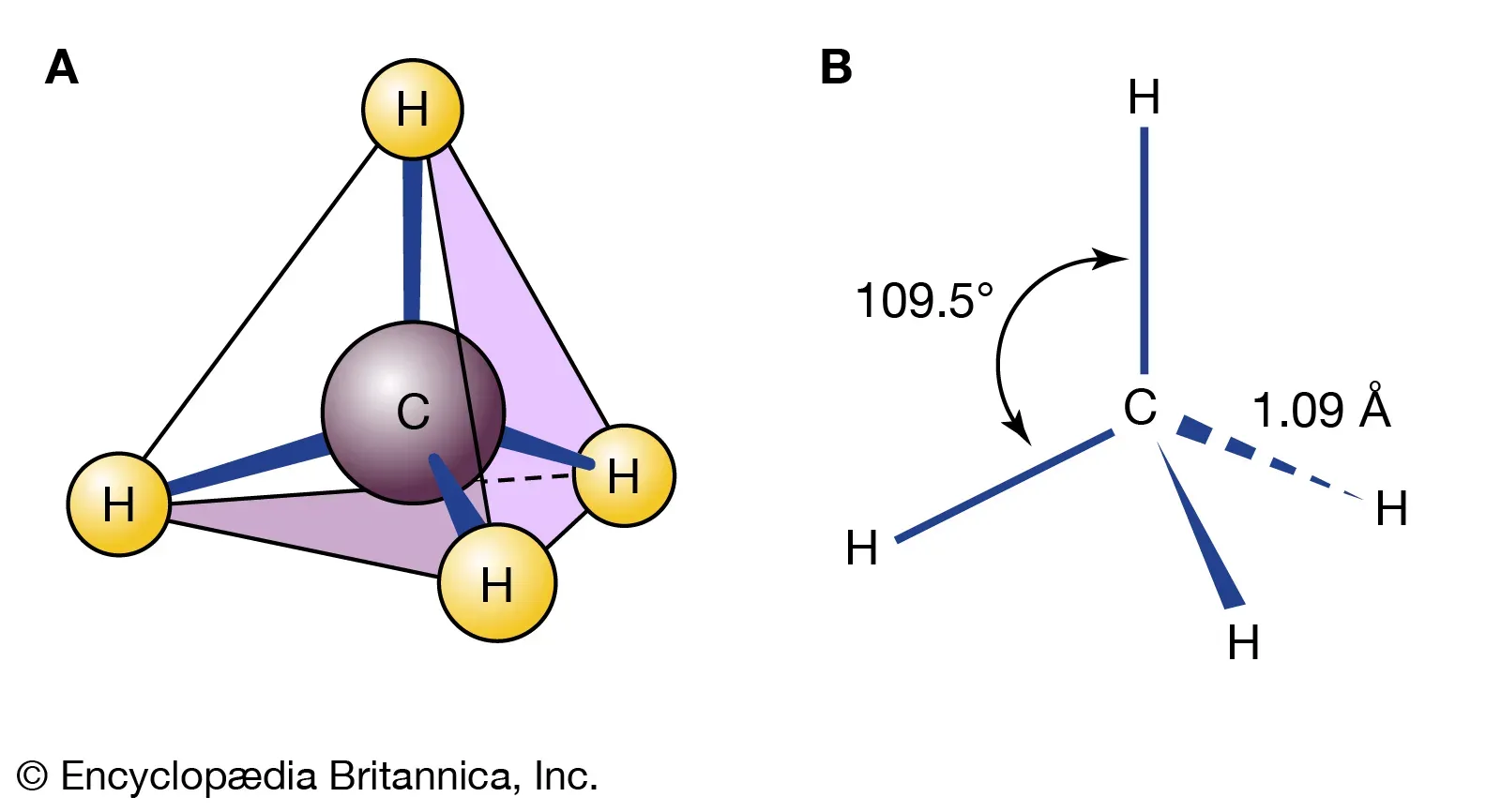
Three Bonding Pairs and One Lone Pair ()
Molecules with three bonding pairs and one lone pair have a trigonal pyramidal shape. The bond angle is less than (approximately ).
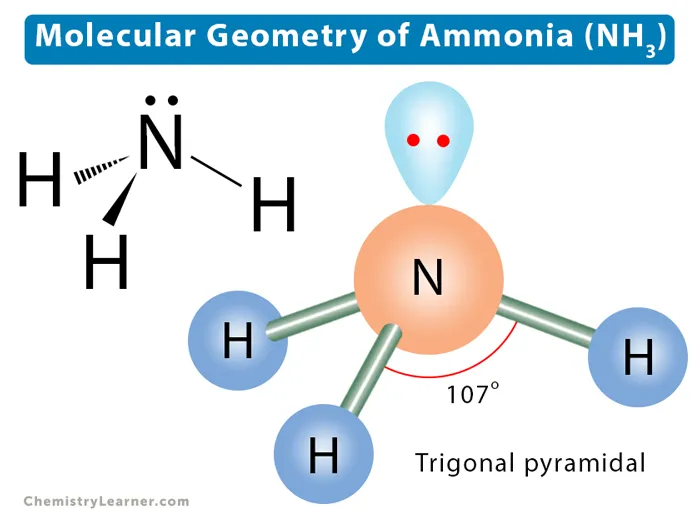
Example: Ammonia ()
- The central nitrogen atom has one lone pair and three bonding pairs.
- The lone pair repels the bonding pairs, reducing the H–N–H bond angle to approximately .
Example: Phosphine ()
- Phosphorus (Group 15) has 5 valence electrons: 3 bond pairs + 1 lone pair → → trigonal pyramidal.
Two Bonding Pairs and Two Lone Pairs ()
Molecules with two bonding pairs and two lone pairs have a bent or V-shaped geometry. The bond angle is significantly less than (approximately ) due to strong LP–LP and LP–BP repulsions.
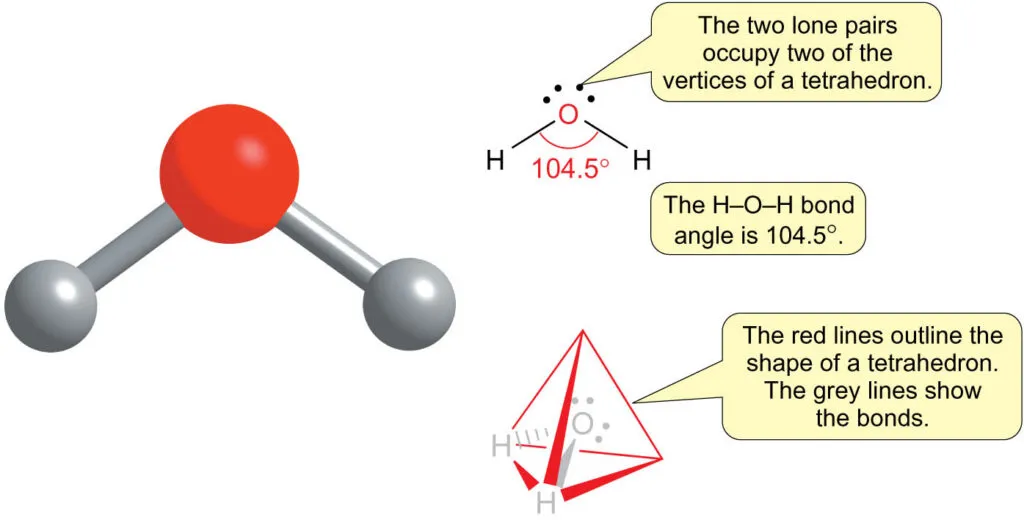
Example: Water ()
- The central oxygen atom has two lone pairs and two bonding pairs.
- The two lone pairs exert a strong repulsive force, compressing the H–O–H bond angle to .
3.5.4 Shapes of Molecules with Five Electron Pairs ()
Molecules with five bonding pairs and no lone pairs have a trigonal bipyramidal geometry. This shape has two distinct positions:
- Axial positions (2): at to the equatorial plane
- Equatorial positions (3): in the central plane at to each other
The axial and equatorial bond angles are not equal ( and respectively).
Example: Phosphorus Pentachloride ()
- Central phosphorus has 5 bonding pairs and no lone pairs → trigonal bipyramidal.
| Summary Table: VSEPR Geometries |
|---|
| Formula Type | Bonding Pairs | Lone Pairs | Shape | Bond Angle |
|---|---|---|---|---|
| 2 | 0 | Linear | ||
| 3 | 0 | Trigonal Planar | ||
| 2 | 1 | Bent | ||
| 4 | 0 | Tetrahedral | ||
| 3 | 1 | Trigonal Pyramidal | ||
| 2 | 2 | Bent | ||
| 5 | 0 | Trigonal Bipyramidal |
Application of VSEPR Theory in Drug Design
The shape and bond angles of molecules are critically important in drug design and pharmacology. This is because:
- Drug molecules must fit precisely into receptor binding sites (active sites) on proteins or enzymes — a concept known as the lock-and-key model.
- Even a small change in molecular geometry (e.g., a different bond angle or lone pair arrangement) can prevent a drug from binding to its target receptor.
- VSEPR theory allows chemists to predict and design the 3D shape of drug molecules to optimize their fit with biological targets.
- For example, the bent shape of and the trigonal pyramidal shape of (due to lone pairs) are relevant to how water and amine-containing drugs interact with biological molecules.
This is why understanding molecular geometry is not just theoretical — it has direct applications in the development of medicines.