3.7 Hybridization
Hybridization is the concept of mixing atomic orbitals of different energies and shapes to produce a new set of equivalent orbitals, called hybrid orbitals. These new orbitals have the same energy and shape.
During hybridization, electrons in the ground state are often promoted to an excited state, which increases the number of unpaired electrons available for bonding. The energy required for this electron promotion is compensated by the energy released during bond formation. The type of hybridization depends on the atomic orbitals involved (s, p, d, etc.). This concept is crucial for explaining molecular geometry and bonding properties.
Types of Hybridization
There are several types of hybridization, including , , , , , and . We will focus on the first three types.
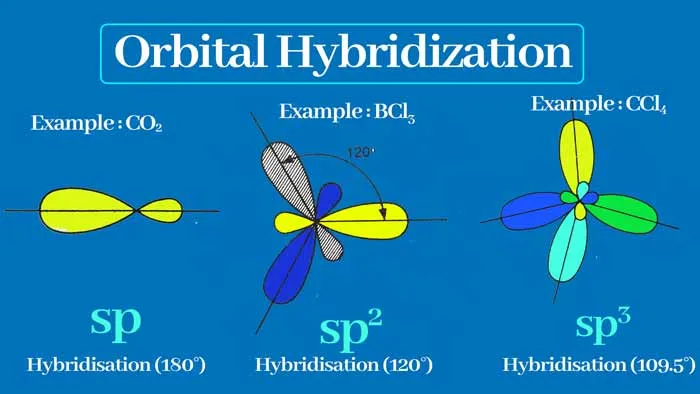
3.7.1 sp Hybridization
- Definition: The intermixing of one s and one p orbital to produce two new, equivalent sp hybrid orbitals.
- Character: Each sp-hybridized orbital has 50% s-character and 50% p-character.
- Geometry: The two sp hybrid orbitals are arranged linearly, with a bond angle of .
- Bonding: sp hybrid orbitals typically form sigma () bonds.
Examples of sp Hybridization
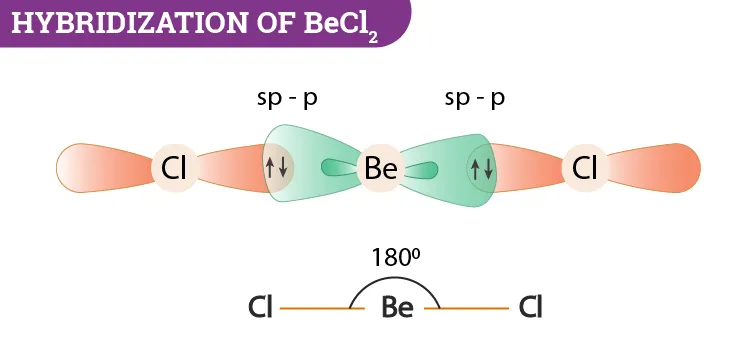
1. Beryllium Chloride ()
- Central Atom: Beryllium (Be)
- Ground State Configuration of Be:
- Excited State: To form two bonds, one electron is promoted to an empty orbital.
- Excited State Configuration of Be:
- Hybridization: The and one orbital mix to form two sp hybrid orbitals.
- Bonding: Each sp hybrid orbital of Beryllium overlaps with a p orbital of a Chlorine atom to form two Be-Cl sigma bonds.
- Resulting Shape: The molecule is linear with a Cl-Be-Cl bond angle of .
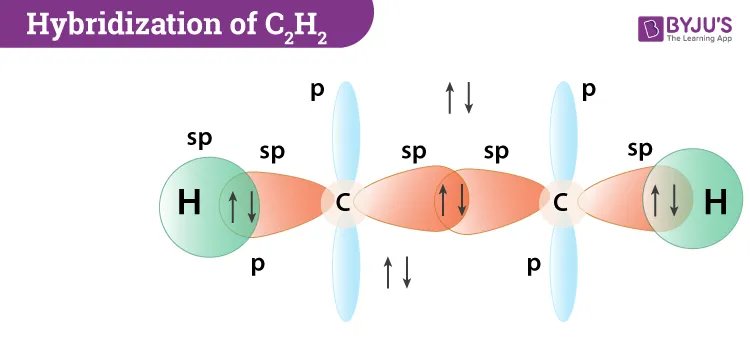
2. Acetylene ()
- Central Atom: Carbon (C)
- Ground State Configuration of C:
- Excited State: To form four bonds, a electron is promoted to the empty orbital.
- Excited State Configuration of C:
- Hybridization: Each carbon atom undergoes sp hybridization, mixing its and one orbital to form two linear sp hybrid orbitals. Two p orbitals ( and ) remain unhybridized on each carbon.
- Bonding:
- One sp orbital from each carbon overlaps to form a C-C sigma () bond.
- The other sp orbital on each carbon overlaps with the 1s orbital of a hydrogen atom to form two C-H sigma () bonds.
- The two unhybridized p orbitals on each carbon overlap sideways to form two pi () bonds between the carbon atoms.
- Resulting Shape: The molecule is linear with H-C-C and C-C-H bond angles of . The C-C bond is a triple bond (one and two ).
3.7.2 Hybridization
- Definition: The intermixing of one s and two p orbitals to produce three new, equivalent hybrid orbitals.
- Character: Each -hybridized orbital has 33.3% s-character and 66.7% p-character.
- Geometry: The three hybrid orbitals are arranged in a trigonal planar geometry, with bond angles of .
Examples of Hybridization
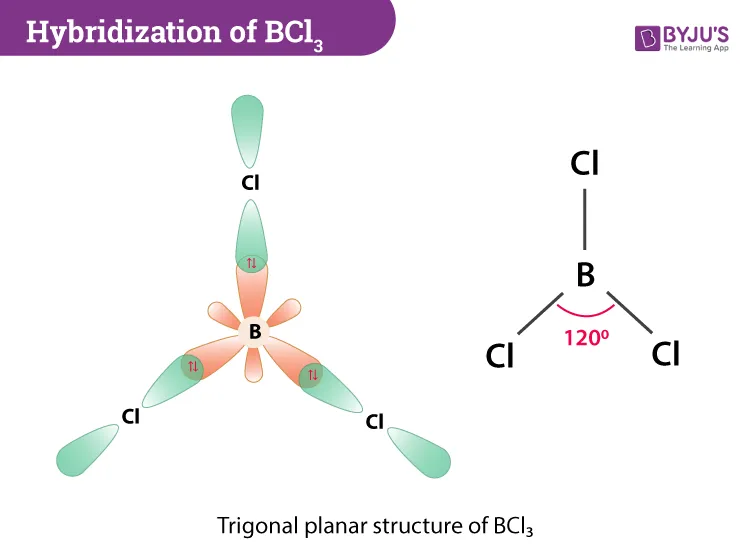
1. Boron Trichloride ()
- Central Atom: Boron (B)
- Ground State Configuration of B:
- Excited State: A electron is promoted to a orbital to make three unpaired electrons available for bonding.
- Excited State Configuration of B:
- Hybridization: The orbital and two orbitals mix to form three hybrid orbitals.
- Bonding: The three hybrid orbitals of Boron each overlap with a p orbital from a Chlorine atom, forming three B-Cl sigma () bonds.
- Resulting Shape: The molecule is trigonal planar with Cl-B-Cl bond angles of .
2. Ethylene ()
- Central Atom: Carbon (C)
- Excited State Configuration of C:
- Hybridization: Each carbon atom undergoes hybridization, mixing its and two orbitals. This leaves one unhybridized orbital on each carbon.
- Bonding:
- One orbital from each carbon overlaps to form a C-C sigma () bond.
- The remaining two orbitals on each carbon overlap with 1s orbitals of hydrogen atoms, forming four C-H sigma () bonds.
- The unhybridized p orbitals on each carbon overlap sideways to form one pi () bond.
- Resulting Shape: The molecule is trigonal planar around each carbon, with H-C-H and H-C-C bond angles of approximately . All six atoms lie in the same plane. The C-C bond is a double bond (one and one ).
3.7.3 Hybridization
- Definition: The intermixing of one s and three p orbitals to produce four new, equivalent hybrid orbitals.
- Character: Each -hybridized orbital has 25% s-character and 75% p-character.
- Geometry: The four hybrid orbitals are arranged in a tetrahedral geometry, with bond angles of (when no lone pairs are present).
Examples of Hybridization
1. Methane ()
- Central Atom: Carbon (C)
- Ground State Configuration of C:
- Excited State: A electron is promoted to the empty orbital.
- Excited State Configuration of C:
- Hybridization: The orbital and three orbitals mix to form four hybrid orbitals.
- Bonding: Each of the four hybrid orbitals overlaps with the 1s orbital of a hydrogen atom, forming four C-H sigma () bonds.
- Resulting Shape: The molecule is tetrahedral with H-C-H bond angles of .
2. Ammonia ()
- Central Atom: Nitrogen (N)
- Ground State Configuration of N:
- Hybridization: Nitrogen undergoes hybridization, forming four hybrid orbitals. Three of these orbitals form N-H sigma bonds with hydrogen atoms; the fourth contains a lone pair of electrons.
- Resulting Shape: Due to the lone pair, the molecular geometry is trigonal pyramidal with H-N-H bond angles of approximately (slightly less than due to lone pair repulsion).
3. Water ()
- Central Atom: Oxygen (O)
- Ground State Configuration of O:
- Hybridization: Oxygen undergoes hybridization, forming four hybrid orbitals. Two of these orbitals form O-H sigma bonds with hydrogen atoms; the remaining two contain lone pairs.
- Resulting Shape: Due to two lone pairs, the molecular geometry is bent (V-shaped) with H-O-H bond angles of approximately (reduced further from due to two lone pairs).
Summary Table
| Hybridization | Orbitals Mixed | No. of Hybrid Orbitals | Geometry | Bond Angle | Example |
|---|---|---|---|---|---|
| 2 | Linear | , | |||
| 3 | Trigonal Planar | , | |||
| 4 | Tetrahedral | , , |