3.8 Co-ordinate Covalent Bond
A coordinate covalent bond (also called a dative covalent bond) is a special type of covalent bond in which one atom provides both electrons for the shared pair.
For a dative covalent bond to form, two conditions must be met:
- One atom must have a lone pair of electrons to donate (Donor).
- The second atom must have an unfilled (empty) orbital to accept the lone pair, making it an electron-deficient atom or molecule (Acceptor).
Examples of Coordinate Covalent Bonds
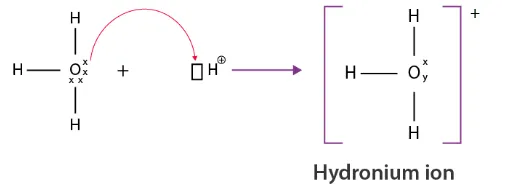
1. Hydronium Ion ()
A coordinate covalent bond is formed when a water molecule () donates one of its lone pairs to an electron-deficient hydrogen ion (, a proton).
The oxygen atom in water provides both electrons to form a new O-H bond, resulting in the hydronium ion.
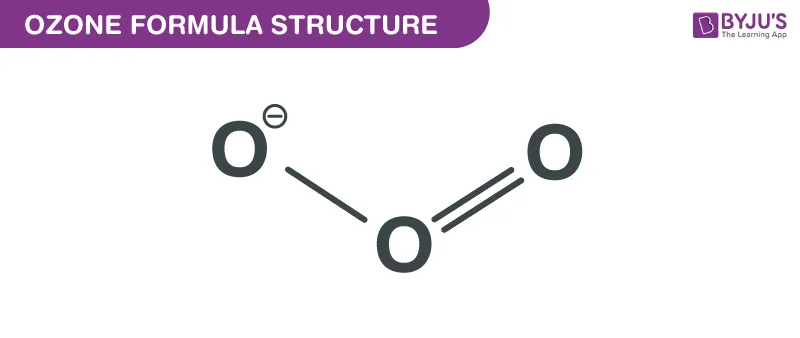
2. Ozone ()
In the ozone molecule, the central oxygen atom forms a double bond with one oxygen atom and a coordinate covalent bond with the other. The central oxygen donates one of its lone pairs to the third oxygen atom.
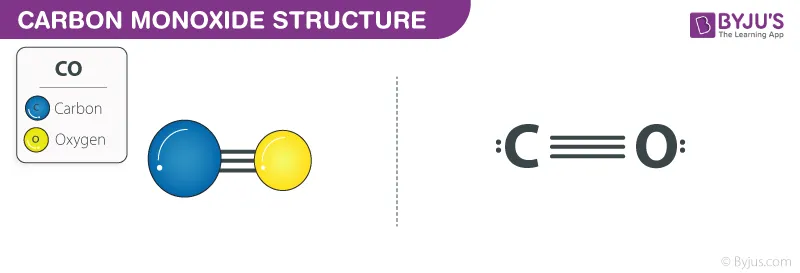
3. Carbon Monoxide ()
In a carbon monoxide molecule, after forming a double bond, the oxygen atom donates a lone pair to the carbon atom to help it achieve a stable octet. This forms a coordinate covalent bond.
Concept Assessment Exercise 3.3
Question 1
Draw dot-and-cross diagrams to show the formation of a coordinate bond between:
i. Boron trichloride () and ammonia () to form the compound .
ii. Phosphine () and a hydrogen ion () to form the ion .
Answer 1:
i. Formation of :
In ammonia (), the nitrogen atom has one lone pair of electrons. In boron trichloride (), the boron atom has an empty p-orbital and is electron-deficient (only 6 valence electrons). Nitrogen donates its lone pair to boron, forming a coordinate covalent bond.
Dot-and-cross representation:
ii. Formation of :
In phosphine (), the phosphorus atom has one lone pair of electrons. The hydrogen ion () has an empty 1s orbital. Phosphorus donates its lone pair to the hydrogen ion to form the phosphonium ion.
Dot-and-cross representation:
Question 2
Draw the displayed formulae of the products formed in Question 1. Show the coordinate bond by an arrow.
Answer 2:
i. : The arrow points from the electron donor (nitrogen) to the electron acceptor (boron).
ii. : The arrow points from the electron donor (phosphorus) to the electron acceptor (hydrogen).