3.9 Intermolecular Forces (Forces Between Molecules)
Intermolecular forces (IMFs) are the attractive or repulsive forces that exist between molecules, particularly covalent molecules. These forces are significantly weaker than the intramolecular forces (like covalent or ionic bonds) that hold atoms together within a molecule. The physical properties of substances, such as melting point, boiling point, and density, are largely determined by the strength of their intermolecular forces.
Key Principle (SLO C-11-B-47): Ionic, covalent, and metallic bonds are always stronger than intermolecular forces. IMFs act between molecules; intramolecular bonds act within molecules.
3.9.1 Types of Intermolecular Forces
There are three primary types of intermolecular forces:
- Permanent Dipole-Dipole Forces
- Instantaneous Dipole-Induced Dipole Forces (London Dispersion Forces / van der Waals forces)
- Hydrogen Bonding
1. Permanent Dipole-Dipole Forces
These forces occur between polar molecules. A molecule is polar if it has a net dipole moment, which arises from an unequal sharing of electrons due to differences in electronegativity. This creates a permanent separation of charge, where one end of the molecule has a partial positive charge () and the other end has a partial negative charge ().
- Example: In hydrogen chloride (), chlorine is more electronegative than hydrogen, creating a dipole: . The positive end of one molecule is attracted to the negative end of a neighboring molecule.
- Non-Polar Molecules: Some molecules contain polar bonds but are non-polar overall because their symmetrical shape causes the individual bond dipoles to cancel out. A key example is carbon tetrachloride ().
- Effect on Properties: The stronger the dipole-dipole forces, the more energy is required to separate the molecules. This leads to higher melting points, boiling points, heats of vaporization, and heats of sublimation.
2. Instantaneous Dipole-Induced Dipole Forces (London Dispersion Forces)
These are the weakest type of intermolecular forces and are present between all molecules, including non-polar ones.
- Mechanism: Electrons in an atom or molecule are in constant, random motion. At any given moment, the electron distribution can be uneven, creating a temporary, instantaneous dipole. This temporary dipole can then induce a temporary dipole in a neighboring molecule, resulting in a weak, short-lived attraction between them.
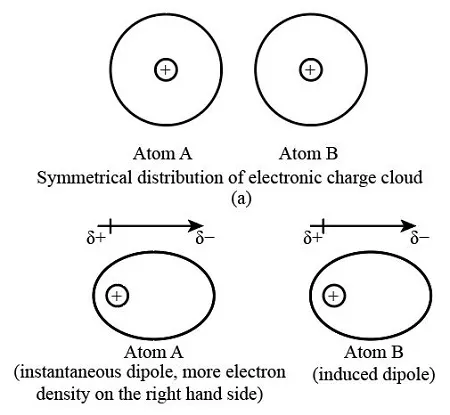
- Factors Affecting Strength: The strength of London Dispersion Forces increases with:
- Molecular size/mass: Larger molecules have more electrons and a larger surface area.
- Number of electrons / Polarizability: A greater number of electrons leads to a more polarizable electron cloud, making it easier to form temporary dipoles.
- Molecular shape: Long, linear molecules have greater surface area for contact than compact, spherical molecules of the same mass.
Example: Boiling Points of Alkanes
As the size of alkane molecules increases, the van der Waals forces become stronger, leading to higher boiling points.
| Name | Molecular Formula | Boiling Point (°C) |
|---|---|---|
| Methane | -164 | |
| Ethane | -89 | |
| Propane | -42 | |
| Butane | -1 | |
| Table 3.5: Boiling point of first four members of alkane. |
For instance, propane (, ) has a higher boiling point than ethane (, ) because its larger size and greater number of electrons result in stronger instantaneous dipole-induced dipole forces.
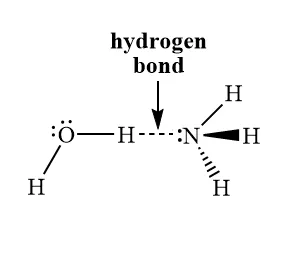
3. Hydrogen Bonding
This is a special, unusually strong type of dipole-dipole interaction. It is not a true chemical bond.
- Conditions: Hydrogen bonding occurs when a hydrogen atom is covalently bonded to a highly electronegative atom with lone pairs of electrons, specifically Nitrogen (N), Oxygen (O), or Fluorine (F).
- Mechanism: The highly electronegative atom pulls the bonding electrons away from the hydrogen atom, leaving a highly exposed, partially positive hydrogen nucleus (). This exposed proton is then strongly attracted to a lone pair of electrons on a neighbouring N, O, or F atom.
- Effect on Properties:
- High Boiling Points: Substances with hydrogen bonding, like water (), have significantly higher boiling points than similar molecules without it.
- Solubility: Covalent compounds that can form hydrogen bonds with water (e.g., ammonia, ) tend to be soluble in water.
3.9.2 Peculiar Behaviour of Water Due to Hydrogen Bonding
Ice is Less Dense Than Water
Normally, the solid state of a substance is denser than its liquid state. Water is a notable exception due to hydrogen bonding.
- In ice, water molecules arrange themselves into a rigid, three-dimensional crystalline lattice.
- Each oxygen atom is tetrahedrally surrounded by four hydrogen atoms, forming a network held together by relatively long hydrogen bonds.
- This creates an open structure with significant empty space — more empty space than exists in liquid water.
- In liquid water, hydrogen bonds are constantly breaking and reforming, allowing molecules to pack more closely together.
- As a result, ice is less dense than liquid water, which is why ice floats. This property is vital for aquatic life, as the floating ice layer insulates the water below.
High Boiling Point of Water
Water (, ) has a much higher boiling point (100°C) than would be expected for such a small molecule. For comparison, hydrogen sulfide (, ) boils at only . This anomalously high boiling point is because water molecules are held together by strong hydrogen bonds (), requiring significantly more energy to vaporize.
Solubility of Ammonia in Water
Ammonia () is highly soluble in water because it can form hydrogen bonds with water molecules: This ability to hydrogen bond with water makes many nitrogen- and oxygen-containing compounds (such as alcohols, amines, and sugars) soluble in water.
3.9.3 Comparison of Intermolecular Force Strengths
| Force Type | Molecules Affected | Relative Strength |
|---|---|---|
| London Dispersion Forces | All molecules | Weakest |
| Permanent Dipole-Dipole | Polar molecules | Moderate |
| Hydrogen Bonding | Molecules with N-H, O-H, or H-F | Strongest IMF |
All intermolecular forces are weaker than intramolecular bonds (ionic, covalent, metallic). This is why molecular substances generally have lower melting and boiling points than ionic or metallic substances.