3.10 Molecular Orbital Theory
The Molecular Orbital (MO) Theory, proposed by Hund and Mullikan in 1932, describes chemical bonding through the combination of atomic orbitals to form molecular orbitals that are characteristic of the entire molecule. When atomic orbitals combine, they lose their individual identities. This process, known as the Linear Combination of Atomic Orbitals (LCAO), results in the formation of new molecular orbitals with different energy levels.
- Bonding Molecular Orbital (BMO): A lower-energy, stable orbital formed from the constructive interference of atomic orbitals.
- Antibonding Molecular Orbital (ABMO): A higher-energy, unstable orbital formed from the destructive interference of atomic orbitals.
MO theory can effectively explain the magnetic properties of molecules:
- Paramagnetic: Molecules with one or more unpaired electrons in their molecular orbitals. They are attracted by a magnetic field.
- Diamagnetic: Molecules with no unpaired electrons. They are repelled by a magnetic field.
3.10.1 Bonding and Antibonding Molecular Orbitals
Bonding Molecular Orbital (BMO)
- Formed by the constructive overlap (addition) of atomic orbitals.
- Characterized by high electron density in the region between the two nuclei, which strengthens the bond and increases stability.
- Electrons in BMOs contribute to the attraction between atoms.
- Designated as sigma () and pi () orbitals.
Antibonding Molecular Orbital (ABMO)
- Formed by the destructive overlap (subtraction) of atomic orbitals.
- Characterized by zero electron density (a nodal plane) between the two nuclei.
- Electron density is concentrated on the opposite sides of the nuclei, leading to repulsion and decreased stability.
- Electrons in ABMOs contribute to repulsion between atoms.
- Designated with an asterisk, as sigma-star () and pi-star () orbitals.
Fundamental Rules:
- The number of molecular orbitals formed is equal to the number of atomic orbitals combined.
- Electrons in molecular orbitals are influenced by all nuclei in the molecule.
- The filling of electrons into molecular orbitals follows established principles:
- Aufbau Principle (fill lowest energy levels first)
- Pauli's Exclusion Principle (max two electrons per orbital, with opposite spins)
- Hund's Rule (fill degenerate orbitals singly before pairing)
Rules of Electronic Configuration→
Types of Overlapping
-
Head-on Approach (Linear Overlapping):
- Occurs with the combination of s-s, s-p, and orbitals.
- Forms sigma () molecular orbitals. For example, two s atomic orbitals combine to form one low-energy (BMO) and one high-energy (ABMO).
-
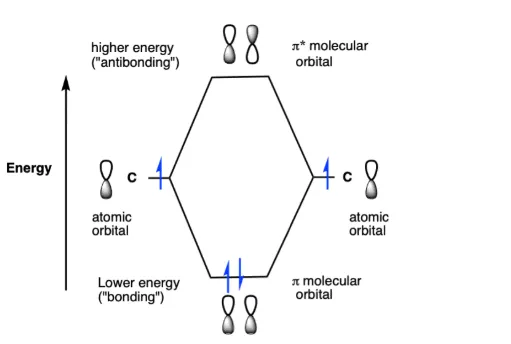
Sideways Approach (Parallel Overlapping):
- Occurs between and orbitals.
- Forms pi () molecular orbitals. For example, two atomic orbitals combine to form one low-energy (BMO) and one high-energy (ABMO).
When the three 2p atomic orbitals () of one atom overlap with those of another, six molecular orbitals are formed (three bonding and three anti-bonding).
3.10.2 Bond Order (No. of Bonds)
Bond order is defined as half the difference between the number of electrons in bonding molecular orbitals and the number of electrons in antibonding molecular orbitals. It represents the number of chemical bonds between two atoms.
- If the bond order is zero, the molecule is unstable and does not exist.
- A positive bond order indicates that the molecule is stable and can exist.
Example: Hydrogen Molecule ()
- Number of electrons in Bonding MOs = 2
- Number of electrons in Antibonding MOs = 0
- Calculation: This indicates a single bond between the two hydrogen atoms.
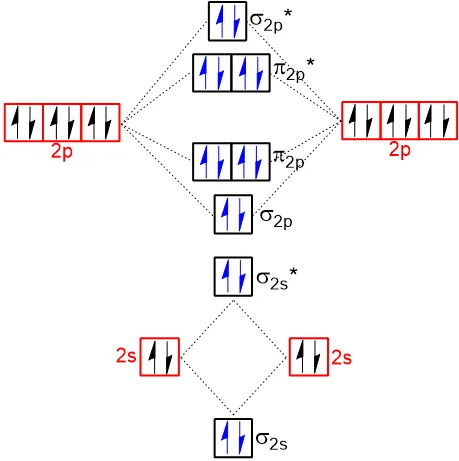
3.10.3 Relative Energies of the Molecular Orbitals
The relative energies of molecular orbitals, determined by spectroscopy, differ for lighter and heavier diatomic molecules of the second period.
a) For Heavier Molecules (, and their ions):
The increasing order of energy is:
b) For Lighter Molecules ():
These molecules show a slightly different order due to s-p mixing, where the energy difference between the 2s and 2p atomic orbitals is small. This mixing raises the energy of the orbital above the orbitals.
Molecular Orbital Diagrams of Homo Nuclear Diatomic Molecules
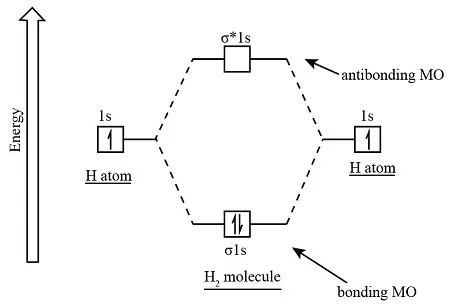
1. Hydrogen Molecule ()
- Total Electrons: 2
- Atomic Configuration (H):
- MO Configuration:
- Bond Order:
- Magnetic Character: Diamagnetic (no unpaired electrons).
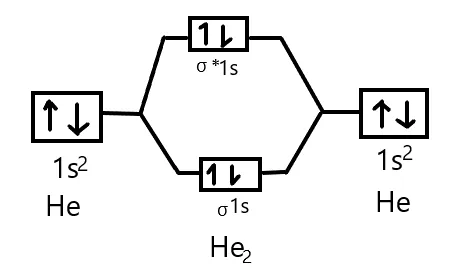
2. Helium Molecule (Hypothetical, )
- Total Electrons: 4
- Atomic Configuration (He):
- MO Configuration:
- Bond Order:
- Magnetic Character: Diamagnetic.
- Conclusion: Since the bond order is zero, the molecule is unstable and does not exist. Helium exists as mono-atomic atoms.
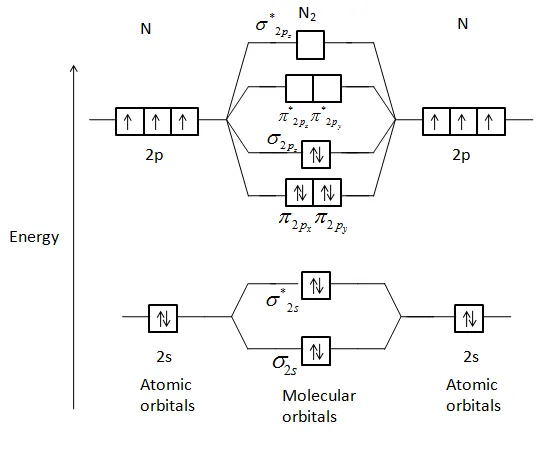
3. Nitrogen Molecule ()
- Total Electrons: 14
- Atomic Configuration (N):
- MO Configuration:
- Bond Order:
- Magnetic Character: Diamagnetic.
- Properties: The bond order of 3 indicates a very stable triple bond (). It has a very high bond energy () and a short bond length.
4. Oxygen Molecule ()
- Total Electrons: 16
- Atomic Configuration (O):
- MO Configuration:
- Bond Order:
Paramagnetic Nature of Oxygen
A key success of MO theory is its ability to explain why oxygen is paramagnetic. The MO diagram for shows two unpaired electrons, one in the orbital and another in the orbital. These unpaired electrons give its magnetic properties. This observation is not explained by the Valence Bond Theory.
- Properties: The bond order of 2 indicates a stable double bond (), with a high bond energy () and a bond length of .
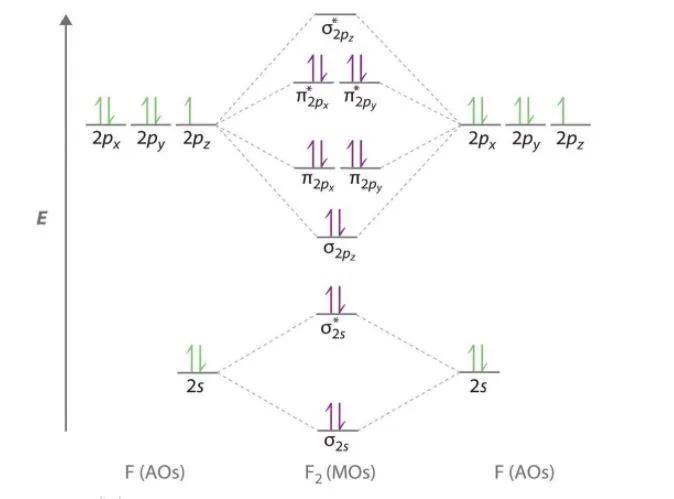
5. Fluorine Molecule ()
- Total Electrons: 18
- Atomic Configuration (F):
- MO Configuration:
- Bond Order:
- Magnetic Character: Diamagnetic.
- Properties: The bond order of 1 indicates a single bond (). The bond energy is relatively low () and the bond distance is longer () compared to and .