2.5 Shapes of Orbitals
An orbital is the region in three-dimensional space around a nucleus where the probability of finding an electron is maximum. Different types of orbitals have distinct shapes, which are determined by the quantum numbers.
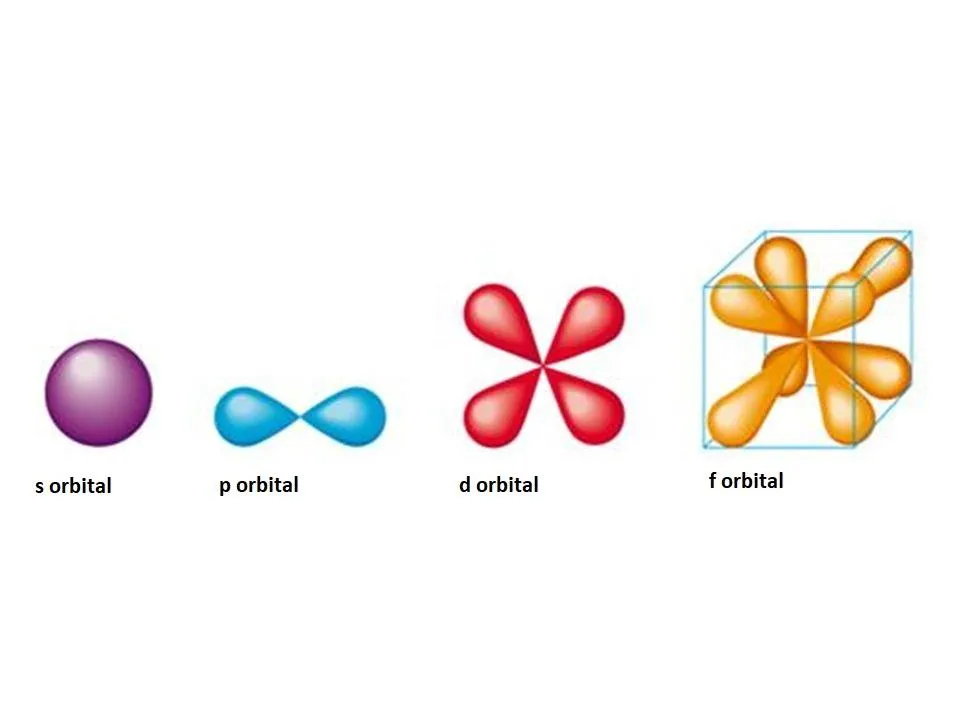
| Orbital Type | Shape Description |
|---|---|
| s-orbital | Spherical |
| p-orbital | Dumbbell |
| d-orbital | Cloverleaf (mostly) |
| f-orbital | Complex / Multi-lobed |
s-Orbital
The s-orbital is spherically symmetrical. Its electron density is highest at the nucleus and decreases with increasing distance from the nucleus. As the principal quantum number () increases, the size of the s-orbital increases, but the spherical shape is retained.
p-Orbital
The p-orbital is dumbbell shaped, consisting of two lobes of equal size separated by a nodal plane passing through the nucleus. There are three p-orbitals (, , ) oriented along the three mutually perpendicular axes.
d-Orbital
The d-orbitals are generally cloverleaf shaped with four lobes, except for the orbital. The five d-orbitals are , , , , and .
f-Orbital
The f-orbitals have more complex, multi-lobed shapes. There are seven f-orbitals.
Free Radicals
A free radical is an atom or molecule that possesses one or more unpaired electrons in its outer shell.
Reactivity
The presence of an unpaired electron makes free radicals highly reactive and capable of initiating immediate chemical reactions. They are often referred to as activated atoms or molecules. This high reactivity is due to their tendency to pair up their unpaired electron.
Rules of Electronic Configuration→
Electronic Configuration
The electronic configuration of a free radical is the same as that of its corresponding neutral atom.
Notation
Free radicals are denoted by placing a dot on the atomic or molecular symbol to represent the unpaired electron.
Examples:
- Chlorine free radical:
- Methyl free radical:
- Bromine free radical:
For instance, the electronic configuration of a chlorine free radical () is identical to that of a neutral chlorine atom ().