2.2 Quantum Numbers
Quantum numbers are a set of four numbers used to describe the location and energy of an electron within an atom. This mathematical approach provides a complete address for each electron, explaining its position within the electronic cloud and its properties. The energy of an electron is primarily determined by the first two quantum numbers.
1. Principal Quantum Number (n)
The Principal Quantum Number, denoted by n, corresponds to the main energy level or shell of an electron, similar to the shells in Bohr's atomic model.
-
Possible Values: It can have any positive integer value.
-
Significance:
- Energy and Size: A larger value of indicates a higher energy level and a larger orbital (the electron is, on average, farther from the nucleus).
- Maximum Electrons: The maximum number of electrons in a shell is . For example: holds 2, holds 8, holds 18 electrons.
- Periodic Table: The value of for the outermost electrons corresponds to the period number in the periodic table.
- Atomic Spectra: It originated from explaining the distinct lines in the emission spectrum of hydrogen, where each line corresponds to an electron transitioning between different shells ( values).
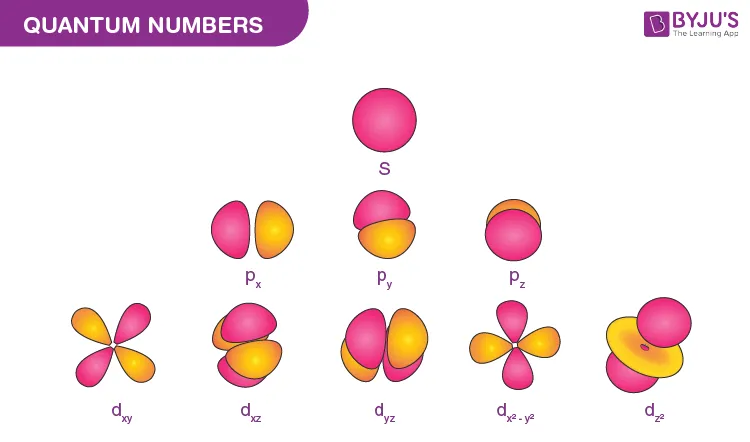
2. Azimuthal Quantum Number (l)
The Azimuthal Quantum Number, also known as the orbital angular momentum quantum number, is denoted by l. It defines the shape of an orbital and specifies the subshell within a principal energy level.
-
Possible Values: The value of depends on . It can range from 0 to .
-
Subshell Notation: Each value of corresponds to a specific subshell, identified by a letter:
- s orbital (spherical)
- p orbital (dumbbell-shaped)
- d orbital (complex shapes)
- f orbital (more complex shapes)
-
Significance:
- Orbital Shape: It describes the three-dimensional shape of the region where an electron is most likely to be found.
- Electrons per Subshell: The maximum electrons in a subshell : s holds 2, p holds 6, d holds 10, f holds 14.
- Periodic Table: The s, p, d, and f subshells correspond to the blocks of the periodic table.
- Atomic Spectra: The existence of subshells explains the fine lines observed in atomic spectra.
3. Magnetic Quantum Number ()
The Magnetic Quantum Number, denoted by , describes the orientation of an orbital in space. Its existence explains the splitting of spectral lines when an atom is placed in a magnetic field (the Zeeman effect).
-
Possible Values: The value of depends on . It can range from to , including 0.
-
Number of Orbitals: The total number of orbitals in a subshell .
-
Significance (Orbital Orientation):
- s-orbital (): . There is only one s-orbital, which is spherically symmetrical.
- p-orbital (): . There are three p-orbitals, oriented along the x, y, and z axes (). These orbitals are degenerate (same energy).
- d-orbital (): . There are five degenerate d-orbitals ().
- f-orbital (): . There are seven degenerate f-orbitals.
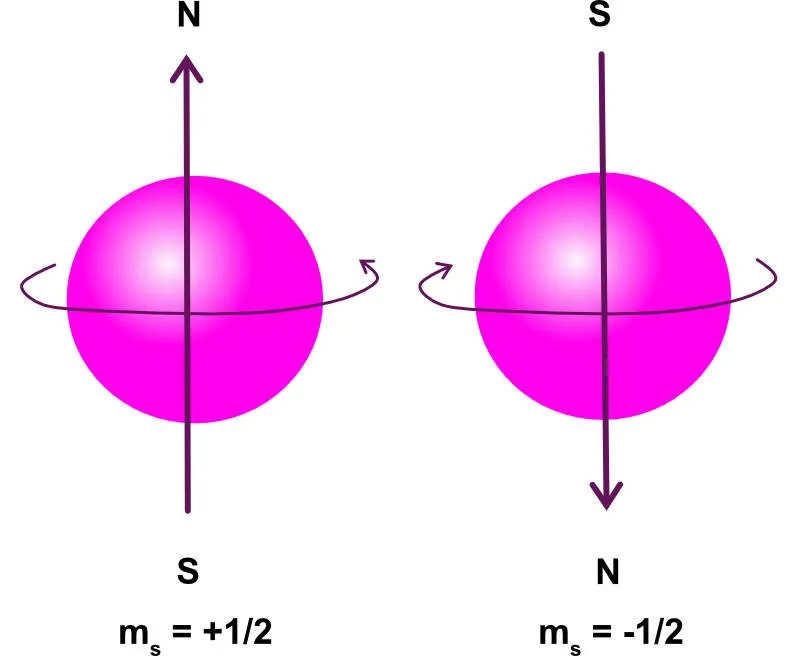
4. Spin Quantum Number ()
The Spin Quantum Number, denoted by , describes the intrinsic angular momentum of an electron, which behaves as if the electron were spinning on its axis. This spin creates a small magnetic field.
-
Possible Values: An electron can only have one of two possible spin states.
-
Significance:
- Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers. Therefore, a single orbital (defined by , , and ) can hold a maximum of two electrons, and they must have opposite spins.
- Notation: A spin-up electron () is represented by , and a spin-down electron () by .
Summary of Quantum Numbers and Orbitals
The table below shows the relationship between quantum numbers and the number of electrons that can be accommodated in each shell.
| Principal Shell (n) | Shell Name | Azimuthal (l) Values | Subshell | Magnetic () Values | Orbitals per Subshell | Electrons per Subshell | Total Electrons in Shell () |
|---|---|---|---|---|---|---|---|
| 1 | K | 0 | 1s | 0 | 1 | 2 | 2 |
| 2 | L | 0 | 2s | 0 | 1 | 2 | 8 |
| 1 | 2p | -1, 0, +1 | 3 | 6 | |||
| 3 | M | 0 | 3s | 0 | 1 | 2 | 18 |
| 1 | 3p | -1, 0, +1 | 3 | 6 | |||
| 2 | 3d | -2, -1, 0, +1, +2 | 5 | 10 | |||
| 4 | N | 0 | 4s | 0 | 1 | 2 | 32 |
| 1 | 4p | -1, 0, +1 | 3 | 6 | |||
| 2 | 4d | -2, -1, 0, +1, +2 | 5 | 10 | |||
| 3 | 4f | -3, -2, -1, 0, +1, +2, +3 | 7 | 14 |
Applications: Quantum Numbers and Electronic Configuration
The four quantum numbers together provide a complete description of every electron in an atom. They are the foundation for writing electronic configurations.
Rules Derived from Quantum Numbers
- Pauli Exclusion Principle (from ): Each orbital holds at most 2 electrons with opposite spins.
- Aufbau Principle (from and ): Orbitals are filled in order of increasing energy, determined by the rule.
- Hund's Rule (from ): Within a subshell, electrons occupy separate orbitals singly before pairing.
Worked Example
For the electron in the 3rd shell, 2nd subshell, 2nd orbital, spin-up: This describes one of the electrons in the subshell.
Quantum Numbers and the Periodic Table
| Quantum Number | Determines | Periodic Table Connection |
|---|---|---|
| Shell / energy level | Period number | |
| Subshell / orbital shape | Block (s, p, d, f) | |
| Orbital orientation | Number of elements per block | |
| Electron spin | Max 2 electrons per orbital |