2.1 Brief History of Atomic Models
This section covers the evolution of atomic theory, the discovery of sub-atomic particles, atomic and mass numbers, behaviour of particles in electric fields, and periodic trends in atomic and ionic radius.
Evolution of Atomic Theory
The concept of the atom has evolved over centuries:
-
Democritus (~430 BCE): Proposed that matter is made of tiny, indivisible particles called atomos (Greek: 'uncuttable'). This was a philosophical idea with no experimental basis and was largely ignored for ~2000 years, overshadowed by Aristotle's view of continuous matter.
-
John Dalton (1808): Transformed the atom from a philosophical concept into a scientific one by basing it on experimental evidence and the laws of chemical combination:
- Law of Conservation of Mass — atoms are neither created nor destroyed in reactions.
- Law of Definite Proportions — atoms of different elements combine in fixed ratios.
- Dalton also proposed that all atoms of a given element are identical in mass and properties. This was later disproved by the discovery of isotopes.
-
Niels Bohr's Model: Introduced quantized energy levels (electronic shells) with fixed energy. It successfully calculated the radius of the hydrogen atom and explained the hydrogen emission spectrum. However, it failed to explain:
- Fine spectrum: Spectral lines split into finer lines under high resolution.
- Stark Effect: Splitting of spectral lines in an electric field.
- Zeeman Effect: Splitting of spectral lines in a magnetic field.
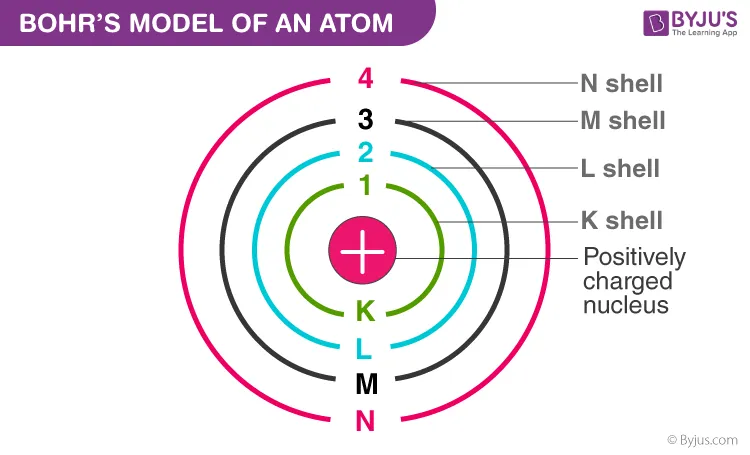
These limitations, along with Heisenberg's Uncertainty Principle, led to the quantum mechanical model involving subshells and orbitals. For details, refer to Quantum Numbers→.
2.1.1 Sub-Atomic Particles
Atoms are composed of three primary sub-atomic particles:
| Particle | Mass (kg) | Charge (C) | Relative Mass (amu) | Relative Charge |
|---|---|---|---|---|
| Proton | ~1 | +1 | ||
| Neutron | 0 | ~1 | 0 | |
| Electron | 1/1836 | −1 |
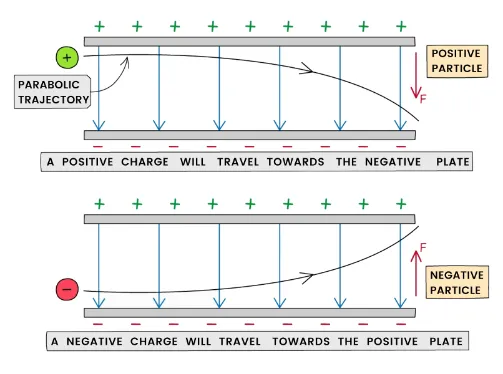
- Electron: Negatively charged; discovered by J.J. Thomson. Deflected towards the positive plate in an electric field.
- Proton: Positively charged. Deflected towards the negative plate. It is 1836 times heavier than an electron.
- Neutron: Electrically neutral; slightly more massive than a proton. Undeflected in electric and magnetic fields.
2.1.2 Behaviour of Sub-Atomic Particles in an Electric Field
When a beam of sub-atomic particles passes through a uniform electric field:
- Electrons (negative charge) → deflected towards the positive plate
- Protons (positive charge) → deflected towards the negative plate
- Neutrons (no charge) → pass through undeflected
The degree of curvature depends on the particle's charge, mass, and velocity. Electrons curve more than protons because they have a much smaller mass.
2.1.3 Atomic Number and Mass Number
-
Atomic Number (Z): The number of protons in the nucleus. Also called the proton number. In a neutral atom:
-
Mass Number (A): The total number of protons + neutrons in the nucleus. Also called the nucleon number. where = number of neutrons.
Calculating Electrons in Ions
- Cation (positive ion): electrons = − (magnitude of charge)
- Example: (): electrons =
- Anion (negative ion): electrons = + (magnitude of charge)
- Example: (): electrons =
2.1.4 Atomic and Ionic Radius
Atomic Radius: The average distance from the nucleus to the outermost electron shell. Since the electron cloud has no sharp boundary, this is an average measure of atomic size.
Periodic Trends in Atomic Radius
1. Across a Period (Left → Right):
- Atomic radius decreases.
- Reason: Nuclear charge (number of protons) increases, pulling electrons closer to the nucleus. The shielding effect from inner electrons remains roughly constant.
- Exception: In transition metals, the trend is less regular because electrons are added to inner d-orbitals, which increases shielding.
2. Down a Group (Top → Bottom):
- Atomic radius increases.
- Reason: Each successive period adds a new electron shell, placing outermost electrons further from the nucleus. Increased shielding from inner electrons also reduces the effective nuclear charge felt by outer electrons.
Ionic Radius
- Cations are smaller than their parent atoms — removing electrons reduces electron-electron repulsion and the remaining electrons are pulled closer by the same nuclear charge.
- Anions are larger than their parent atoms — adding electrons increases electron-electron repulsion, expanding the electron cloud.