2.3 Rules of Electronic Configuration
This section outlines the fundamental principles that govern how electrons are arranged in atomic orbitals. These rules ensure that the electron configuration of an atom corresponds to its lowest possible energy state, also known as the ground state.
Aufbau Principle
The Aufbau principle (from the German Aufbau, meaning "building up") states that electrons are distributed into atomic orbitals in order of increasing energy. Orbitals with lower energy are filled before those with higher energy.
The energies of orbitals are determined by quantum mechanical calculations. The general order for filling orbitals is as follows:
This sequence can be visualized using a diagram that helps remember the filling order.
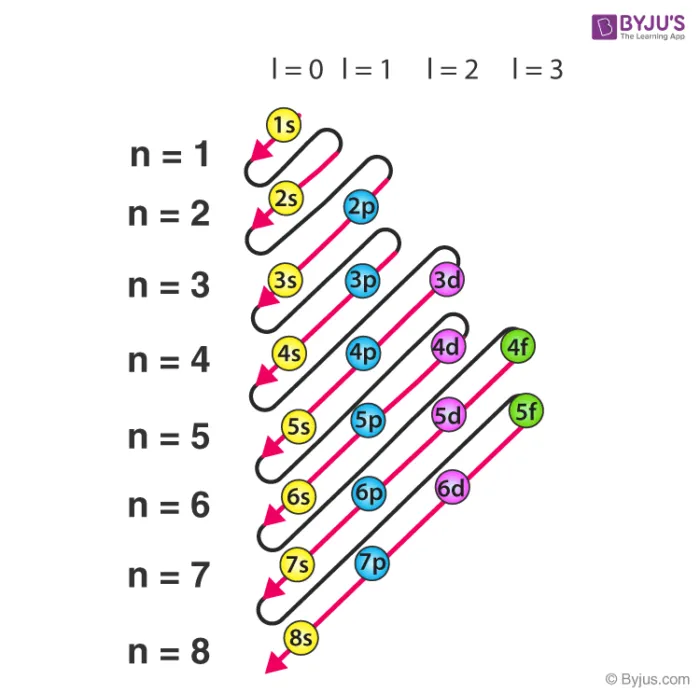
The Rule (Madelung's Rule)
The rule is a guideline used to determine the energy order of orbitals, which explains why some higher principal shells () are filled before lower ones.
- Primary Rule: The orbital with a lower value of has lower energy and is filled first.
- Secondary Rule: If two orbitals have the same value, the orbital with the lower principal quantum number () has lower energy.
Example:
- For the 4s orbital:
- For the 3d orbital:
Since , the 4s orbital has lower energy than the 3d orbital and is filled first.
Pauli Exclusion Principle
The Pauli exclusion principle states that no two electrons in an atom can have the same set of all four quantum numbers ().
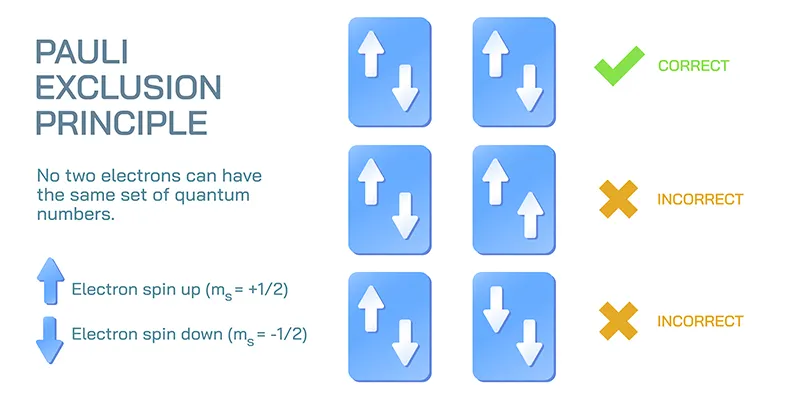
This means that if two electrons occupy the same orbital (i.e., they have the same and values), they must have opposite spins. One electron will have a spin-up state (, denoted by ) and the other will have a spin-down state (, denoted by ).
Example: The two electrons in a orbital have the following quantum numbers:
| Electron A | Electron B | |
|---|---|---|
| Principal () | 3 | 3 |
| Azimuthal () | 1 | 1 |
| Magnetic () | -1 (for ) | -1 (for ) |
| Spin () | +1/2 () | -1/2 () |
This principle implies that any single atomic orbital can hold a maximum of two electrons.
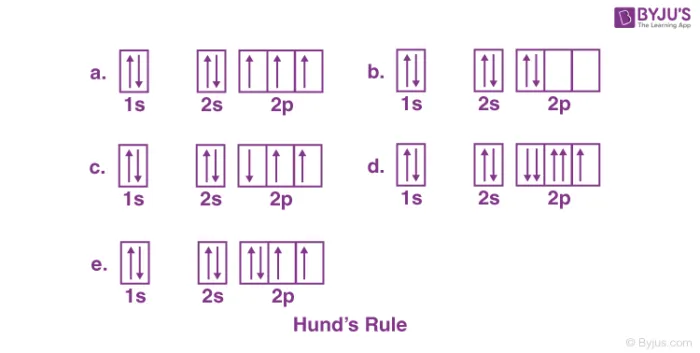
Hund's Rule of Maximum Multiplicity
Hund's rule applies when filling a set of degenerate orbitals (orbitals with the same energy, such as the three p-orbitals or the five d-orbitals).
The rule states that:
- Electrons will first occupy each degenerate orbital singly with parallel spins (all spin-up or all spin-down) before any orbital is doubly occupied.
- Once all degenerate orbitals have one electron, subsequent electrons will pair up with the existing electrons in each orbital, adopting an opposite spin.
Example: Filling the p-orbitals:
- Two electrons in p-orbitals: They will occupy two different orbitals with the same spin.
- Three electrons in p-orbitals: They will occupy all three orbitals singly with the same spin.
Stability Note: This rule is related to the enhanced stability of half-filled and completely filled subshells. This extra stability is evident in the electron configurations of elements like Chromium (Cr) and Copper (Cu), which deviate from the standard Aufbau filling order to achieve a more stable half-filled or fully-filled d-subshell.
- Cr (): Expected ; Actual (half-filled )
- Cu (): Expected ; Actual (fully-filled )
Free Radicals
A free radical is a chemical species (atom, molecule, or ion) that possesses one or more unpaired electrons in its outermost shell.
- Free radicals are formed when a covalent bond breaks homolytically — each atom receives one electron from the shared pair.
- The presence of an unpaired electron makes free radicals highly reactive, as they tend to pair up their electron by reacting with other species.
- Free radicals are denoted by a dot (•) next to the symbol, e.g., , .
Example: Homolytic cleavage of : Each chlorine atom has one unpaired electron and is a free radical.
Worked Examples
Q1: In which orbital of the atom will the 11th electron be placed?
A: The filling order is which accounts for the first electrons. The 11th electron will go into the next available orbital of lowest energy, which is the 3s orbital.
Q2: Place these orbitals in increasing energy order: 5s, 4p, 4s, 3d.
A: Using the rule:
| Orbital | |||
|---|---|---|---|
| 4s | 4 | 0 | 4 |
| 3d | 3 | 2 | 5 |
| 4p | 4 | 1 | 5 |
| 5s | 5 | 0 | 5 |
For orbitals with , the one with lower has lower energy: .
Complete order:
Q3: Which of the following orbitals has greater energy: 5f or 6p?
A:
- 5f:
- 6p:
Since , the 5f orbital has greater energy.
Q4: Which of the following orbitals has greater energy: 4f or 5d?
A:
- 4f:
- 5d:
Both have . Applying the secondary rule, the orbital with lower has lower energy. Since , the 4f orbital has lower energy and the 5d orbital has greater energy.