2.6 Ionization Energy
Ionization Energy (IE) is defined as the minimum energy required to remove the outermost, most loosely bonded electron from an isolated atom in its gaseous state. It is a measure of how strongly an atom holds onto its valence electrons.
General Equations:
The process is endothermic, requiring energy input.
For Sodium (Na):
For Magnesium (Mg):
Note: Atoms must be in the gaseous state to ensure the measurement is not influenced by other energy changes like heat of fusion, bond dissociation energy, or heat of vaporization.
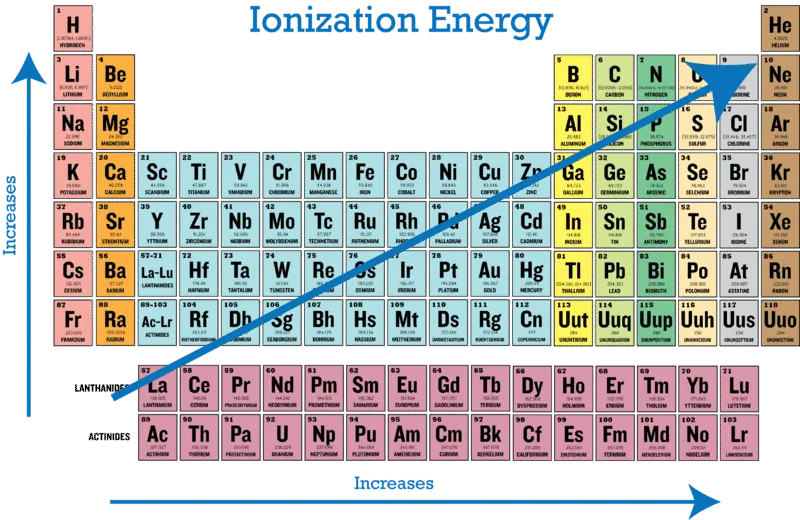
2.6.1 Periodic Trends of Ionization Energy
The ionization energy of elements shows predictable patterns across the periodic table.
Across a Period (Left to Right)
-
Trend: Ionization energy generally increases from left to right across a period.
-
Reasoning:
- Increased Nuclear Charge: The number of protons in the nucleus increases, leading to a stronger electrostatic attraction between the nucleus and the electrons.
- Decreasing Atomic Radius: Electrons are pulled closer to the nucleus, making them more difficult to remove.
- Constant Shielding Effect: For main group elements, the number of inner electron shells remains the same, so the shielding effect is relatively constant and does not counteract the increasing nuclear charge.
-
Anomalies:
-
Group 2 (IIA) to Group 13 (IIIA): There is a slight decrease in IE (e.g., from Be to B). This is because the electron being removed from a Group 13 element is in a higher-energy p-orbital, which is slightly further from the nucleus and more shielded than the s-orbital electrons of the Group 2 element.
-
Group 15 (VA) to Group 16 (VIA): There is a slight decrease in IE (e.g., from N to O). This is due to the stability of the half-filled p-orbital in Group 15 elements. In Group 16, the electron is removed from a p-orbital containing a pair of electrons. The repulsion between these paired electrons (spin-pair repulsion) makes it slightly easier to remove one of them.
-
Down a Group (Top to Bottom)
- Trend: Ionization energy decreases from top to bottom down a group.
- Reasoning:
- Increasing Atomic Radius: The number of electron shells increases, placing the valence electrons further from the nucleus.
- Increased Shielding Effect: The additional inner shells of electrons shield the valence electrons from the full attractive force of the nucleus, reducing the effective nuclear charge () they experience.
2.6.2 Factors Influencing Ionization Energy
Several factors determine the magnitude of an element's ionization energy.
-
Nuclear Charge (): A higher nuclear charge (more protons) results in a stronger attraction for electrons, thus increasing the ionization energy. This is the dominant factor across a period.
-
Atomic Radii: A smaller atomic radius means the valence electrons are closer to the nucleus and held more tightly, which increases the ionization energy.
-
Shielding Effect: Inner-shell electrons "shield" the valence electrons from the full pull of the nucleus. A greater shielding effect makes it easier to remove a valence electron, thus decreasing the ionization energy. This is the dominant factor down a group.
-
Spin-Pair Repulsion: When two electrons occupy the same orbital, they repel each other. This repulsion raises the energy of the electrons, making them slightly easier to remove compared to an electron in a half-filled orbital. This explains the dip in IE from Group 15 to Group 16.
2.6.3 Deduction of Element Position and Configuration from IE Data
Successive ionization energies (the energy to remove a 2nd, 3rd, 4th, etc., electron) provide valuable information about an element's electronic structure and its position in the periodic table.
Deducing Position in the Periodic Table
An element's first ionization energy can help estimate its position relative to other elements.
- Example (Across a Period): If an element 'X' has a first ionization energy () of , it would likely be placed between Sulfur () and Argon ().
- Example (Down a Group): If an element 'Y' has , it could be placed in Group 2 between Beryllium () and Calcium (). This value corresponds to Magnesium (Mg).
Deducing Electronic Configuration (Valence Electrons)
A large jump between successive ionization energies indicates the removal of an electron from a stable, inner electron shell (a core electron). This jump reveals the number of valence electrons.
-
Sodium (Na):
- The enormous jump between and indicates that after one electron is removed, the next electron must be taken from a core shell. This shows Na has one valence electron and belongs to Group 1.
-
Magnesium (Mg):
- The large jump between and shows that the first two electrons are removed from the valence shell, and the third comes from a core shell. This confirms Mg has two valence electrons and belongs to Group 2.
-
Aluminium (Al):
- The large jump between and confirms Al has three valence electrons and belongs to Group 13.
Key Rule: The position of the large jump in successive IE data = number of valence electrons = Group number (for main group elements).