2.7 Mass Spectrometry
Mass spectrometry is an analytical technique used to determine the molecular composition and structure of a sample by measuring the mass-to-charge ratio () of ions. It is widely used in chemistry, biology, and physics.
2.7.1 Principle of Mass Spectrometry
Mass spectrometry operates on a three-step principle:
- Ionization: The sample's neutral atoms or molecules are converted into charged ions.
- Mass Analysis (Separation): The ions are separated based on their mass-to-charge ratio () in a magnetic or electric field.
- Detection: The separated ions are detected, and their relative abundance is measured. This data is used to generate a mass spectrum.
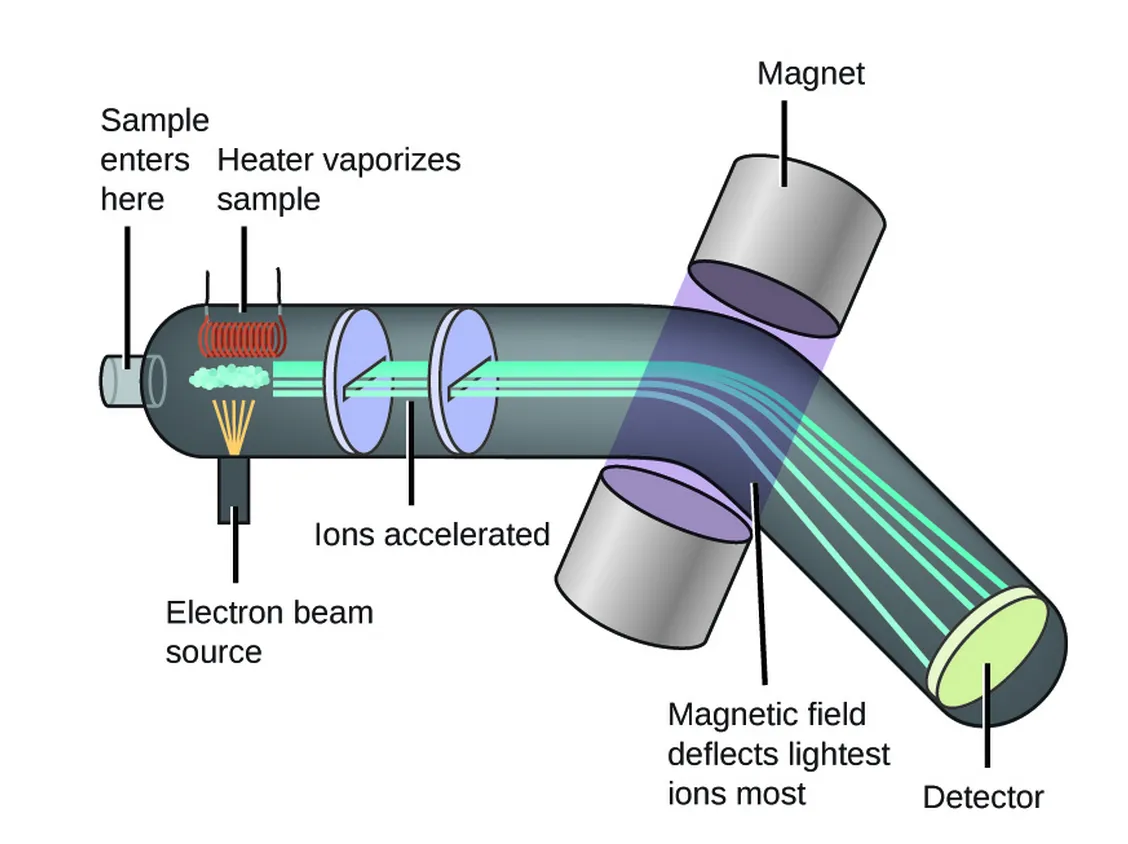
2.7.2 Working of a Mass Spectrometer
The process within a mass spectrometer can be broken down into four stages:
1. Ionization
The sample is introduced into the ion source where it is vaporized and ionized. Vaporization is essential because ions must move freely through the fields.
Common ionization methods include:
- Electron Impact (EI): Bombarding the sample with high-energy electrons.
- Electrospray Ionization (ESI): Introducing the sample to a high-voltage field.
- Matrix-Assisted Laser Desorption/Ionization (MALDI): Using a laser to vaporize and ionize the sample, which is embedded in a matrix.
2. Ion Separation (Mass Analyzer)
The newly formed ions are accelerated by an electric field and directed into the mass analyzer. The analyzer uses magnetic and/or electric fields to separate the ions according to their mass-to-charge () ratio. Lighter ions or ions with a higher charge are deflected more than heavier or less charged ions.
3. Detection
After passing through the analyzer, the separated ions strike a detector. The detector records the number of ions (abundance) for each value.
4. Data Analysis
The detector's signal is processed to create a mass spectrum. This is a plot where:
- The x-axis represents the mass-to-charge () ratio.
- The y-axis represents the relative abundance of the ions.
Each peak in the spectrum corresponds to a specific ion, with its position indicating its and its height indicating its relative abundance.
2.7.3 Applications
Mass spectrometry has a wide range of applications, including:
- Determining the relative abundance of isotopes of an element.
- Identifying unknown compounds by their mass.
- Elucidating the molecular structures of complex molecules.
- Quantifying the amount of specific substances in a sample.
- Studying large biomolecules like proteins and nucleic acids.
Non-Integer Relative Atomic Masses
The atomic masses listed in the periodic table are often not whole numbers. This is because they are a weighted average of the masses of an element's naturally occurring isotopes. Mass spectrometry provides the data (mass and relative abundance of each isotope) needed to calculate this average.
The average atomic mass is calculated using the following formula:
Worked Examples
Calculating the Average Atomic Mass of Chlorine
Chlorine has two naturally occurring isotopes: chlorine-35 and chlorine-37.
-
Write the given values:
- Isotope 1: Chlorine-35, Mass = 35 amu, Relative Abundance = 75.76%
- Isotope 2: Chlorine-37, Mass = 37 amu, Relative Abundance = 24.24%
-
Apply the formula:
-
Show the calculation: Therefore, the relative atomic mass of chlorine is 35.48 amu.
Possible Questions/Answers
Q: Calculate the average atomic mass of Magnesium (Mg) using the data from its mass spectrum (Figure 2.10).
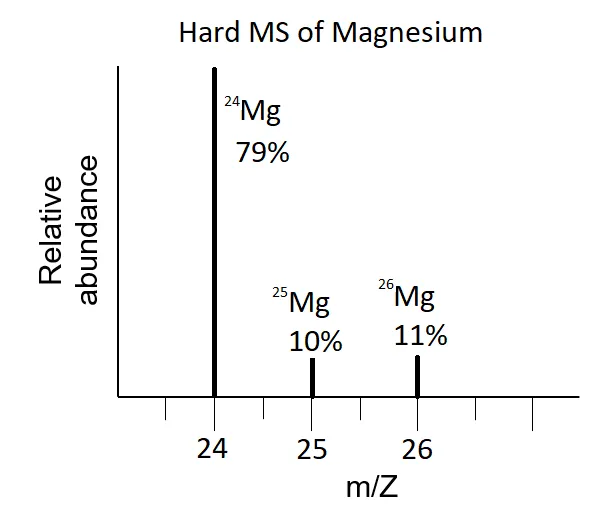
A:
-
Identify isotopes and abundances from the spectrum:
- Magnesium-24: 78.70%
- Magnesium-25: 10.13%
- Magnesium-26: 11.17%
-
Apply the average atomic mass formula: The average atomic mass of Magnesium is 24.32 amu.