10.3 POSITION OF AN ELEMENT IN THE PERIODIC TABLE
10.3 POSITION OF AN ELEMENT IN THE PERIODIC TABLE
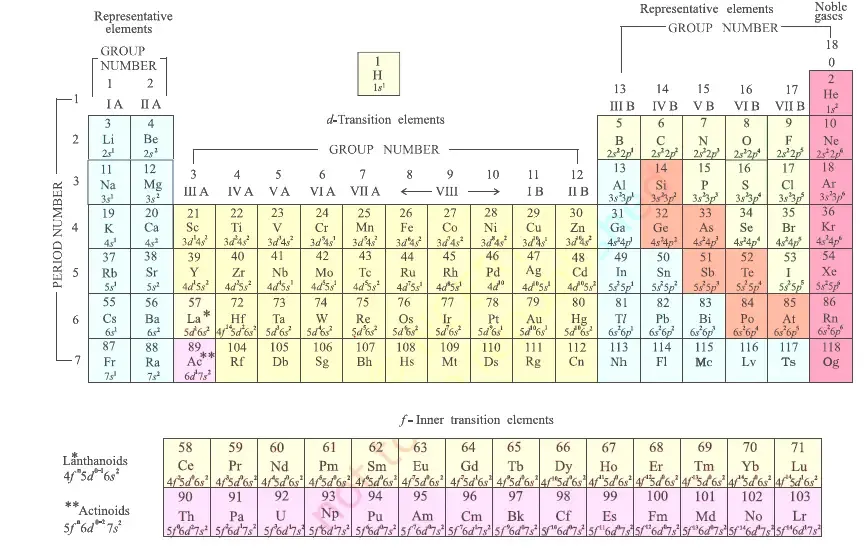
The electronic configuration of an element is the key to determining its position in the periodic table. By analyzing the valence electron configuration, specifically the principal quantum number () and the sub-shell type, you can identify an element's period, group, and block.
Determining the Position of an Element
The location of an element is defined by its period (horizontal row) and group (vertical column).
1. Period Number
The period number corresponds to the principal quantum number (n) of the outermost or valence shell.
- If the valence electrons of an element are in the L shell (), the element belongs to the 2nd period.
- If the valence electrons are in the M shell (), the element belongs to the 3rd period.
2. Group Number
The group number is determined by the number of electrons in the valence shell.
- For s-block elements: The group number is equal to the total number of electrons in the valence shell.
- For p-block elements: The group number is found by adding 10 to the total number of valence electrons.
- For example, if the valence shell configuration is , the total number of valence electrons is . The element belongs to Group 15 (or VA). The calculation is:
- For d-block elements: The group number equals the number of electrons in the subshell plus the number of electrons in the subshell.
- For example, Scandium (): Group = .
Characteristic Properties of Elements in a Group
Chemical periodicity refers to the repeating pattern of elemental properties in the periodic table. Elements within the same group share similar chemical properties because they have analogous valence electron configurations.
- Atomic Size: Gradually increases from top to bottom in a group. Elements lower in the group have larger atomic radii because additional electron shells are added.
- Ionization Energy: Generally decreases from top to bottom in a group. It is easier to remove an electron from elements lower in the group due to increased atomic radius and greater shielding effect. Ionization Energy→
- Electronegativity: Decreases down the groups. Elements further down a group are less electronegative because the nucleus exerts a weaker pull on bonding electrons.
- Metallic Properties: Increase as you move down a group. Elements at the bottom of a group lose electrons more easily, making them more metallic.
- Chemical Reactivity: Elements in the same group exhibit similar chemical reactivity due to having the same number of valence electrons.
Example 10.1: Find the position of the following elements in the periodic table.
(a) Nitrogen (atomic number 7) (b) Oxygen (atomic number 8)
Problem Solving Strategy:
- Write the full electron configuration of the element. Rules of Electronic Configuration→
- Identify the valence shell and its configuration.
- The principal quantum number () of the valence shell gives the period number.
- The total number of electrons in the valence shell (plus 10 for p-block) gives the group number.
Solution:
(a) Nitrogen (N)
- Electronic Configuration:
- Valence Shell Configuration:
- Period Number: The highest value is 2.
- Group Number:
- Total valence electrons = .
- Since it is a p-block element, Group = .
- Conclusion: Nitrogen is in the 2nd period and Group 15 (VA).
(b) Oxygen (O)
- Electronic Configuration:
- Valence Shell Configuration:
- Period Number: The highest value is 2.
- Group Number:
- Total valence electrons = .
- Since it is a p-block element, Group = .
- Conclusion: Oxygen is in the 2nd period and Group 16 (VIA).
Example 10.2: Determine the valence shell electronic configuration.
(a) Phosphorus (P) (b) Neon (Ne)
Problem Solving Strategy:
- Identify the period number to find the principal quantum number () of the valence shell.
- Identify the group number to find the total number of valence electrons.
- Distribute the valence electrons into the sub-shells ( and ) of the valence shell.
Solution:
(a) Phosphorus (P)
- Position: Phosphorus is in Period 3 and Group 15 (VA).
- Valence Shell: Since it is in Period 3, . The valence sub-shells are and .
- Valence Electrons: Since it is in Group 15, it has 5 valence electrons.
- Configuration: The 5 electrons are distributed as follows: 2 in the sub-shell and the remaining 3 in the sub-shell.
- Valence shell electronic configuration:
(b) Neon (Ne)
- Position: Neon is in Period 2 and Group 18 (VIIIA).
- Valence Shell: Since it is in Period 2, . The valence sub-shells are and .
- Valence Electrons: Since it is in Group 18, it has 8 valence electrons.
- Configuration: The 8 electrons are distributed as follows: 2 in the sub-shell and the remaining 6 in the sub-shell.
- Valence shell electronic configuration:
Do You Know?
What Makes Up >99% of the Normal Matter in the Universe?
Hydrogen and Helium are the most abundant elements, accounting for approximately 98% of all matter (roughly 73% hydrogen and 25% helium by mass). All other elements combined make up less than 2% of the universe's normal matter.