10.4 Metals, Non-metals, and Metalloids
The periodic table serves as a fundamental framework for organizing chemical elements. Based on their properties and positions within the table, elements are broadly classified into three main categories: metals, non-metals, and metalloids.
Classification of Elements
Elements are categorized based on their distinct physical and chemical properties, which correspond to their location on the periodic table. A prominent dividing line, often shown as a staircase or leader line, helps distinguish these groups.
1. Metals
- Position: Located on the left side of the periodic table.
- Physical Properties:
- Good conductors of heat and electricity (due to delocalized/free electrons).
- Malleable: Can be hammered or pressed into thin sheets without breaking.
- Ductile: Can be drawn into thin wires.
- Lustrous (shiny) appearance.
- Generally solid at room temperature (exception: mercury, Hg).
- Chemical Behavior: Metals tend to lose electrons to form positive ions (cations). They have low ionization energies, making electron loss easy.
Example: Sodium loses one electron:
2. Non-metals
- Position: Located on the right side of the periodic table (to the right of the leader line). Exception: Hydrogen (H) is a non-metal but is placed at the top left.
- Physical Properties:
- Poor conductors of heat and electricity (insulators). Exception: Graphite (a form of carbon) conducts electricity.
- Brittle: Non-malleable — will break or crumble if hammered.
- Non-ductile: Cannot be drawn into wires.
- Dull appearance (not lustrous).
- Chemical Behavior: Non-metals tend to gain or share electrons to form negative ions (anions) or covalent bonds. They have high ionization energies and high electronegativities.
Example: Chlorine gains one electron:
3. Metalloids
- Position: Located adjacent to the leader line (staircase boundary) that separates metals and non-metals, primarily in the p-block.
- Properties:
- Possess properties intermediate between those of metals and non-metals.
- Also known as semi-metals.
- Many act as semiconductors (e.g., Silicon and Germanium), making them essential in electronics and photovoltaic cells.
- Examples: Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), Tellurium (Te).
The Periodic Table Layout
The visual organization of the periodic table clearly shows the separation of these three categories.
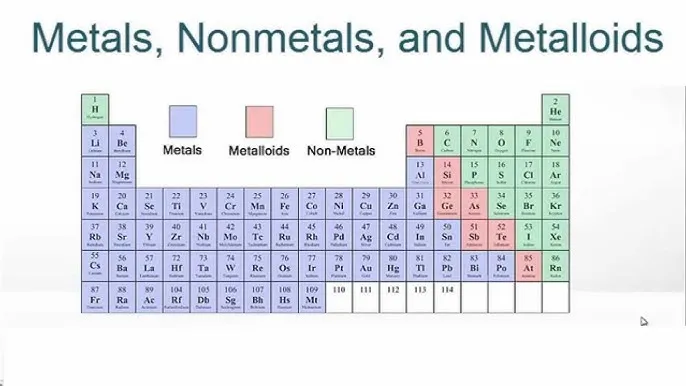
| Category | Position on Periodic Table | Key Properties |
|---|---|---|
| Metals | Left side of the leader line | Good conductors, malleable, ductile, low ionization energy |
| Non-metals | Right side of the leader line | Poor conductors, brittle, non-ductile, high ionization energy |
| Metalloids | Adjacent to the leader line (p-block) | Intermediate properties, semiconductors |
Metallic Bonding — Why Metals Have Unique Properties
The unique properties of metals (conductivity, malleability, ductility) are explained by metallic bonding:
- Metal atoms release their valence electrons into a sea of delocalized electrons that are free to move throughout the metallic lattice.
- Electrical conductivity: The free electrons carry charge through the metal.
- Thermal conductivity: Free electrons transfer kinetic energy rapidly.
- Malleability/Ductility: Metal cations can slide past each other without breaking bonds, since the electron sea maintains cohesion.
This is in contrast to non-metals, where electrons are localized in covalent bonds and cannot move freely.