3.3 Factors Affecting Enzymatic Action Rate
The rate of an enzymatic reaction is determined by measuring the amount of substrate converted or the amount of product formed over a specific period. Several external factors can influence this rate.
Temperature
Temperature affects the kinetic energy of both enzyme and substrate molecules.
-
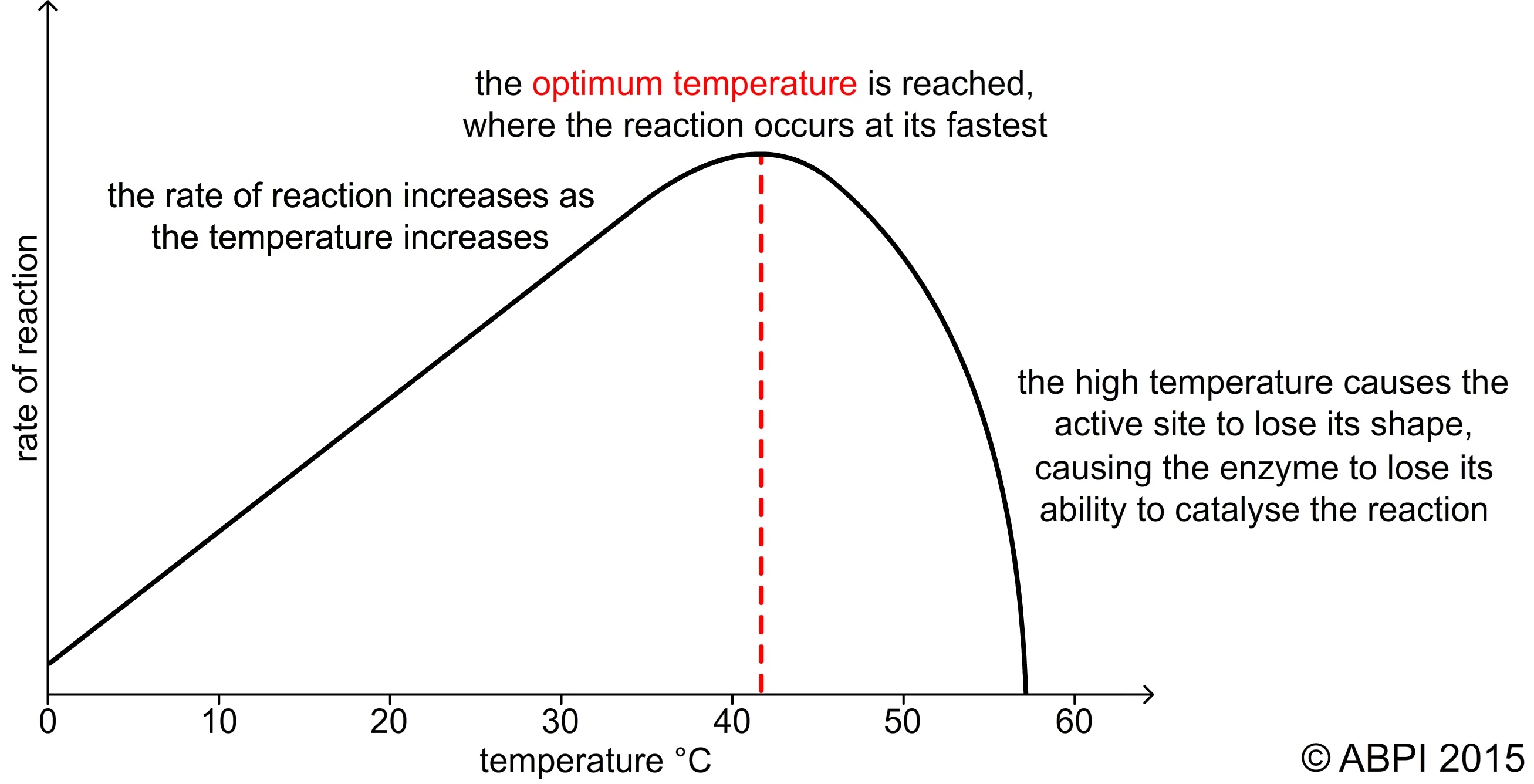
Initial Temperature Increase: As temperature rises, molecules move faster, increasing the frequency of collisions between enzymes and substrates. This leads to a higher rate of reaction.
-
Rule of Thumb: For many biological reactions, a 10°C rise in temperature doubles the reaction rate, up to the optimum.
-
Optimum Temperature: This is the temperature at which the enzyme exhibits maximum activity.
-
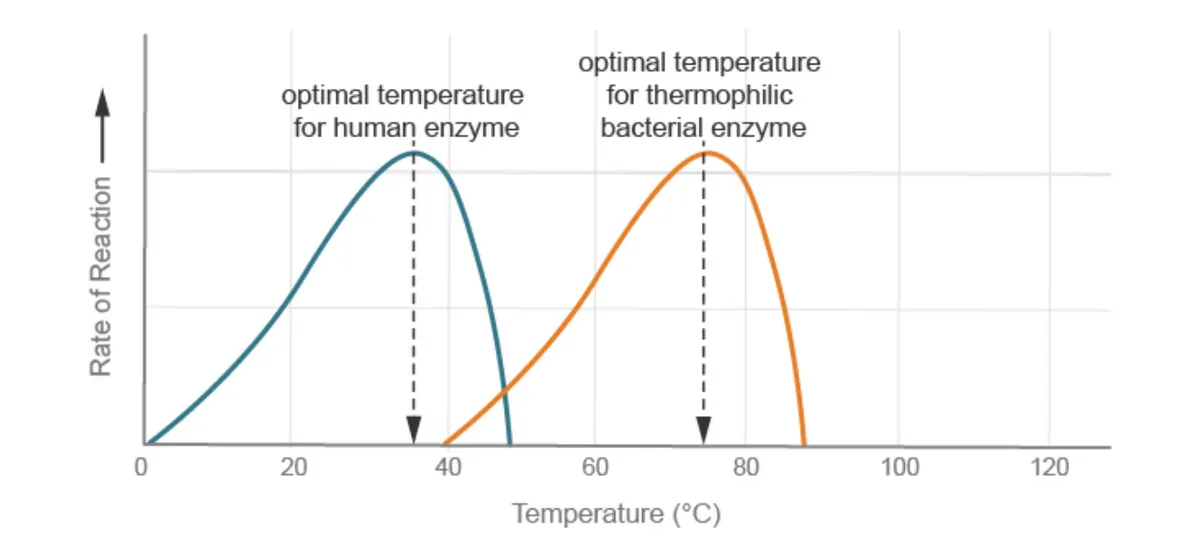
For most human enzymes, the optimum temperature is around 37-38°C.
-
Bacteria living in hot springs (thermophilic bacteria) have much higher optimum temperatures.
-
High Temperatures (Above Optimum):
- Excessive heat disrupts the weak bonds (like hydrogen bonds) that maintain the enzyme's specific three-dimensional (secondary and tertiary) structure.
- This change in shape, particularly of the active site, is called denaturation.
- A denatured enzyme is no longer functional, causing a rapid decrease in the reaction rate.
pH
pH measures the acidity or alkalinity of a solution and significantly impacts enzyme structure and function.
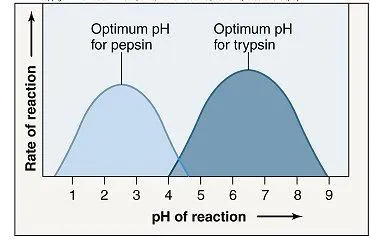
- Optimum pH: Each enzyme has a narrow pH range where it functions most effectively.
- Effect of pH Changes:
- pH affects the ionization (charge) of the amino acid side chains in the enzyme.
- These charges are crucial for maintaining the ionic bonds that stabilize the enzyme's tertiary structure.
- Slight pH changes: Can cause temporary inactivation by altering the charge of the active site.
- Extreme pH changes: Can cause irreversible denaturation by permanently disrupting the enzyme's shape.
Examples of Optimum pH
| Enzyme | Location/Source | Optimum pH | Medium |
|---|---|---|---|
| Pepsin | Stomach | ~2.0 | Highly Acidic |
| Trypsin | Small Intestine | ~8.0 | Alkaline |
| Papain | Papaya | 5.0 - 8.0 | Wide Range (Acidic to Alkaline) |
Enzyme Concentration
The concentration of the enzyme directly influences the overall rate of reaction, assuming other factors are not limiting.
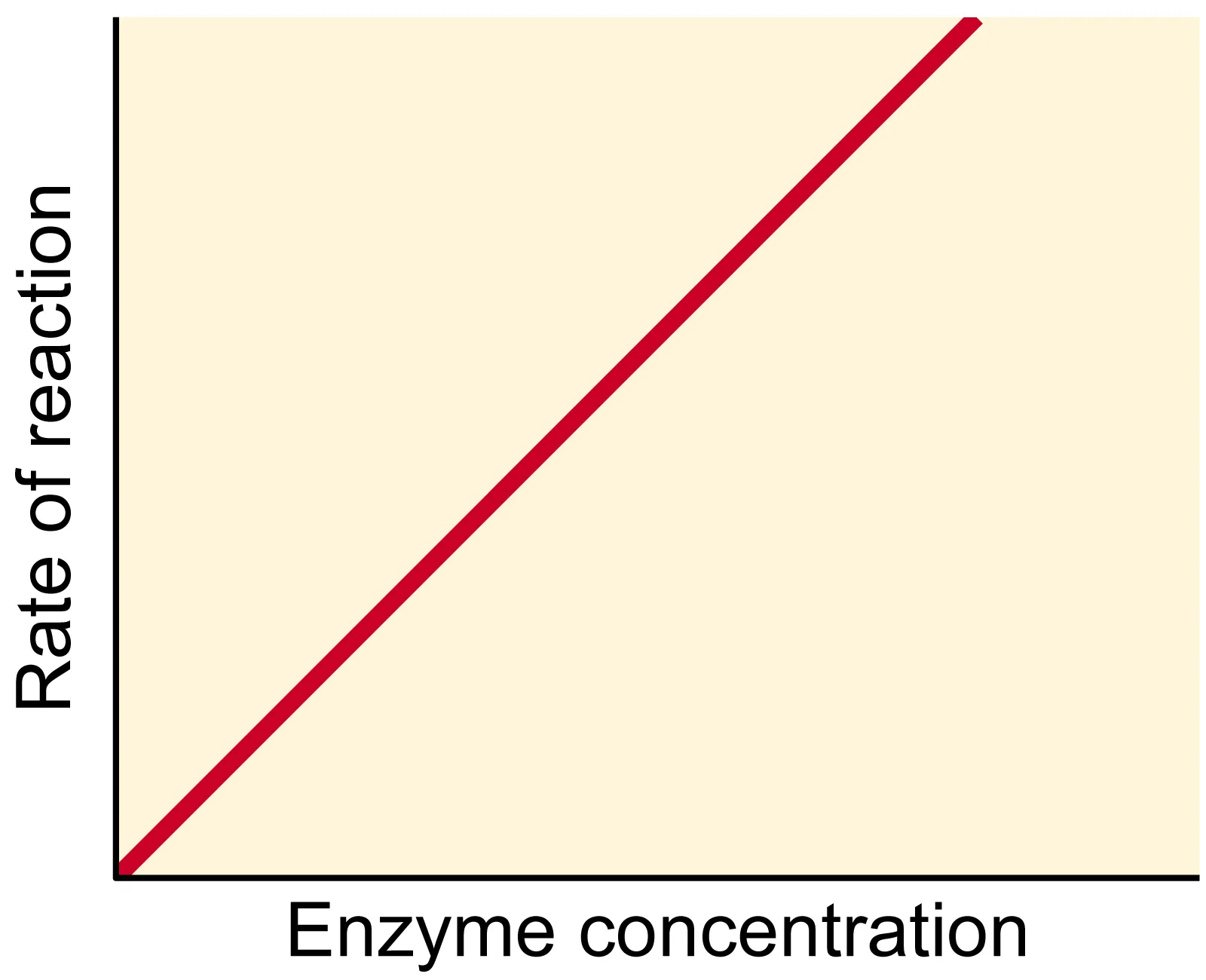
- Relationship: The rate of reaction is directly proportional to the enzyme concentration.
- Condition: This relationship holds true only if the substrate concentration is in excess (unlimited) and factors like temperature and pH are kept constant at their optimal levels.
- Mechanism: Increasing the number of enzyme molecules increases the number of available active sites, leading to more frequent formation of enzyme-substrate complexes and thus a faster reaction rate.
Substrate Concentration
When the enzyme concentration is kept constant, the rate of reaction increases with increasing substrate concentration. However, this increase continues only up to a certain point.
- Saturation Point: At high substrate concentrations, all available active sites of the enzymes are occupied (saturated). At this point, any further increase in substrate concentration will not increase the rate of reaction.
Inhibitors
Inhibitors are substances that decrease the rate of enzymatic reactions by interfering with the active site or changing the enzyme's shape.
- Competitive Inhibitors: These molecules resemble the substrate and compete for the active site.
- Non-competitive Inhibitors: These bind to a site other than the active site (allosteric site), changing the enzyme's conformation so the substrate can no longer bind.
Biological Significance
The sensitivity of enzymes to these factors is crucial for the regulation of metabolic activity within cells. Organisms must maintain stable internal conditions (homeostasis), such as body temperature and pH, to ensure their enzymes function correctly. For more on how cells maintain their environment, see Membrane Transport Mechanism→.