3.2 Mechanism of Enzyme Action
This section details the process by which enzymes catalyze biochemical reactions, focusing on the interaction between the enzyme and its substrate, the models that describe this interaction, and the concept of activation energy.
General Mechanism
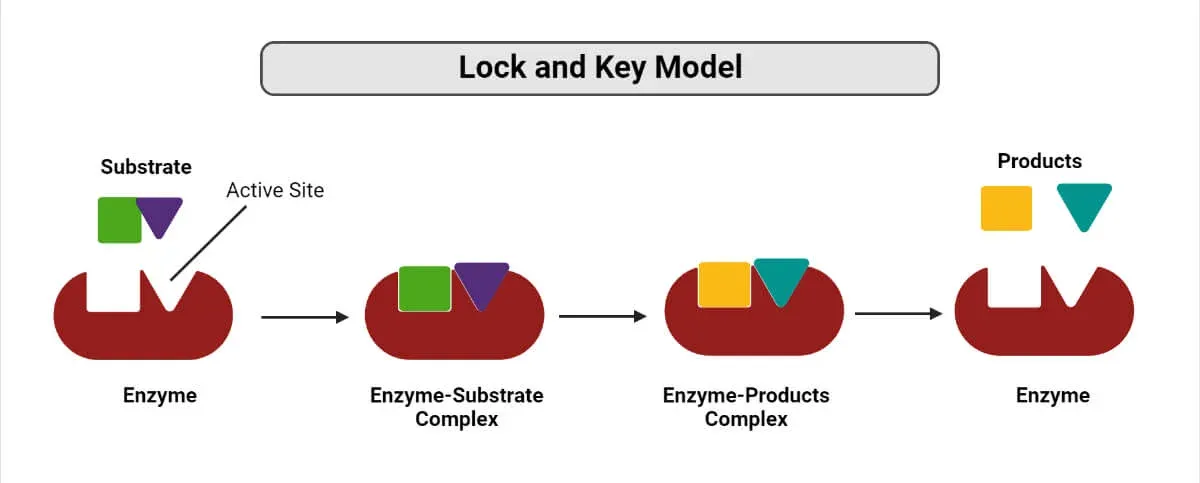
An enzyme-catalyzed reaction follows a specific sequence of events:
-
The substrate (reactant) binds to the active site of the enzyme.
-
This forms a temporary Enzyme-Substrate (ES) complex.
-
While bound, the substrate is converted into the product, forming an Enzyme-Product (EP) complex.
-
The product is released from the active site, leaving the enzyme unchanged and free to catalyze another reaction.
Reaction Pathway:
Models of Enzyme Action
Two primary models explain the interaction between an enzyme's active site and its substrate.
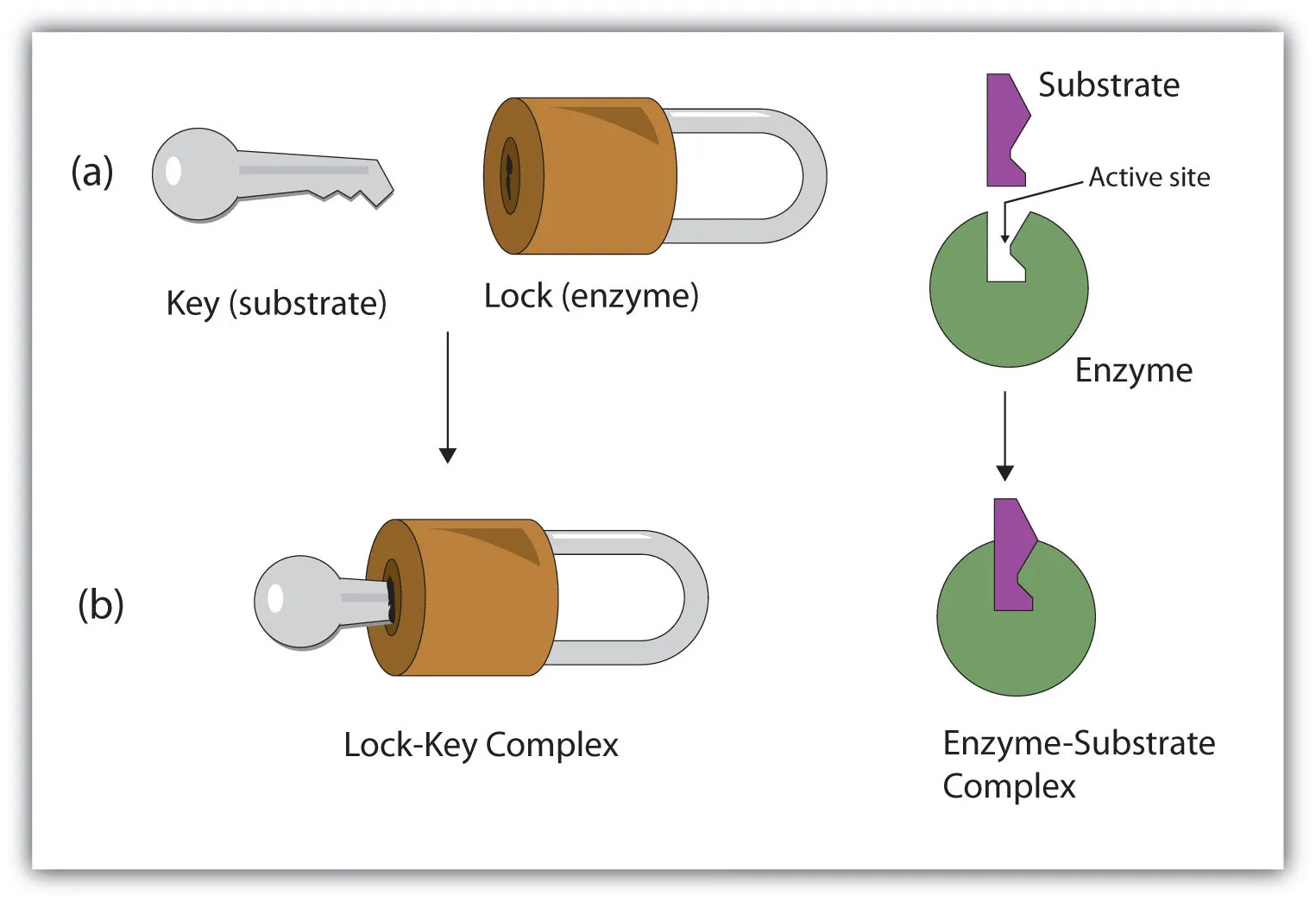
1. Lock and Key Model
-
Proposer: Emil Fischer (1894)
-
Concept: The active site of the enzyme has a rigid, specific shape that is precisely complementary to the shape of its substrate.
-
Analogy: The enzyme is the "lock," and the substrate is the "key." Only the correctly shaped key (substrate) can fit into the lock (enzyme).
-
Specificity: This model explains absolute specificity, where an enzyme can catalyze only one specific reaction on a single substrate.
- Example: The enzyme urease only acts on urea.
-
Enzyme Type: Primarily applies to non-regulatory enzymes like sucrase and maltase.
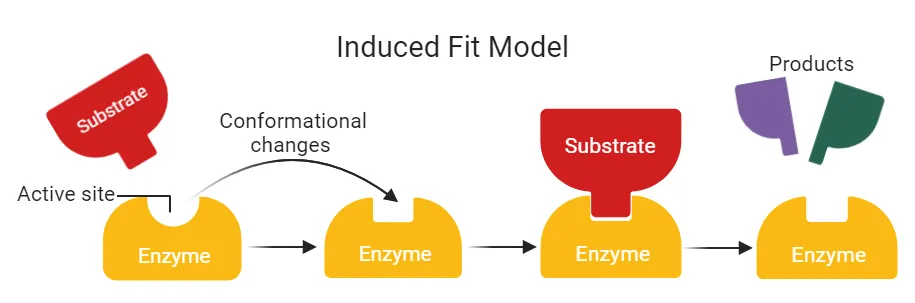
2. Induced Fit Model
-
Proposer: Daniel Koshland (1959)
-
Concept: The active site is flexible, not rigid. The binding of the substrate induces a conformational change in the active site's shape to achieve a more precise fit.
-
Function: This adjustment molds the active site perfectly around the substrate, enhancing the enzyme's catalytic ability. After the reaction, the active site returns to its original shape.
-
Specificity: This model explains why some enzymes can act on a range of related substrates (broader specificity).
-
Enzyme Type: Applies to regulatory or allosteric enzymes.
- Example: Hexokinase and carbonic anhydrase.
Comparison of Models
| Feature | Lock and Key Model | Induced Fit Model |
|---|---|---|
| Proposer | Emil Fischer (1894) | Daniel Koshland (1959) |
| Active Site Nature | Rigid and pre-shaped | Flexible and dynamic |
| Substrate Binding | Substrate fits precisely into the existing active site shape. | Substrate binding induces a conformational change in the active site. |
| Specificity | Explains absolute specificity. | Explains broader specificity for related substrates. |
| Enzyme Type | Non-regulatory enzymes (e.g., sucrase, maltase) | Regulatory/allosteric enzymes (e.g., hexokinase) |
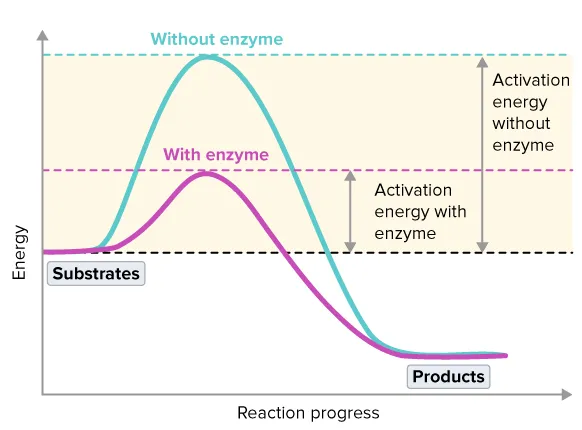
Energy of Activation ()
-
Definition: The Energy of Activation () is the minimum amount of energy required to start a chemical reaction by breaking existing chemical bonds.
-
Enzymes as Catalysts: Enzymes act as biological catalysts by lowering the activation energy required for a reaction.
-
Mechanism: They provide an alternative reaction pathway that requires less energy. The enzyme forms a transitional complex with the substrate (AE complex), which facilitates the conversion to the product.
-
Significance: By lowering , enzymes dramatically increase the rate of chemical reactions, allowing them to occur rapidly at the relatively low and stable temperatures found within living organisms. Importantly, enzymes do not change the equilibrium constant of the reaction.