3.4 Enzyme Inhibition
Enzyme inhibition is the process where a substance, known as an inhibitor, binds to an enzyme and decreases its activity. This is a crucial mechanism for regulating enzyme activity within cells, but it can also be induced by external factors like poisons or drugs.
General Concepts
- Enzyme Inhibition: A process where a molecule, an inhibitor, binds to an enzyme and prevents it from catalyzing its reaction. The substrate may still bind, but it is not converted into a product.
- Inhibitors: Molecules that cause inhibition. Examples include poisons (cyanide), drugs (penicillin, sulpha drugs), antibodies, and anti-metabolites.
Types of Inhibition
There are two primary types of enzyme inhibition: competitive and non-competitive.
1. Competitive Inhibition
This occurs when an inhibitor molecule competes directly with the substrate for the enzyme's active site.
-
Mechanism:
- The competitive inhibitor is structurally similar to the normal substrate.
- It fits into the active site, physically blocking the substrate from binding.
- The enzyme cannot catalyze a reaction with the inhibitor.
-
Reversibility: Competitive inhibition is typically temporary and reversible. The inhibitor binds and unbinds from the active site.
-
Effect of Substrate Concentration: This type of inhibition can be overcome by increasing the substrate concentration. With more substrate molecules available, they are more likely to bind to the active sites than the inhibitor molecules.
-
Example: Malonate
- The enzyme succinate dehydrogenase catalyzes the conversion of succinate to fumarate.
- Malonate is structurally similar to succinate and acts as a competitive inhibitor, blocking the active site.
-
Significance:
- Supports the lock and key hypothesis of enzyme action.
- Demonstrates enzyme specificity.
- Used in medicine, for example, sulphonamides (sulpha drugs) are competitive inhibitors that block a key enzyme in bacteria, preventing their growth.
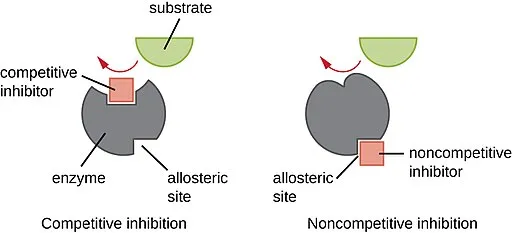
2. Non-Competitive Inhibition
This occurs when an inhibitor binds to the enzyme at a location other than the active site, known as the allosteric site.
- Mechanism:
- The inhibitor's binding to the allosteric site changes the three-dimensional shape of the enzyme.
- This change alters the shape of the active site, so the substrate can no longer bind effectively or, if it does bind, the enzyme cannot catalyze the reaction.
- Reversibility: Non-competitive inhibition can be either reversible or irreversible.
- Reversible: The inhibitor binds temporarily and can detach, allowing the enzyme to regain its function. It prevents the formation of the enzyme-product complex. Feedback inhibition is a key example.
- Irreversible: The inhibitor binds permanently, often through covalent bonds, and denatures the enzyme. The enzyme is permanently destroyed.
- Effect of Substrate Concentration: Increasing the substrate concentration does not overcome non-competitive inhibition because the inhibitor is not competing for the active site.
- Examples of Irreversible Inhibitors:
- Cyanides: Inhibit cytochrome oxidase, an enzyme essential for cellular respiration, by binding to its iron prosthetic group.
- Heavy Metal Ions (, , ): These ions bind to thiol (-SH) groups in the enzyme, breaking the disulfide bridges that maintain the enzyme's tertiary structure and causing denaturation.
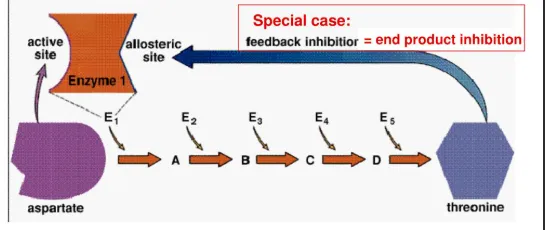
3. Feedback Inhibition
A specific type of reversible, non-competitive inhibition that serves as a natural regulatory mechanism in cells.
- Mechanism: The end product of a metabolic pathway binds to an allosteric site on an enzyme that functions early in the pathway, inhibiting its activity.
- Purpose: This prevents the cell from wasting resources by overproducing a substance it already has in sufficient quantity.
- Example: In the synthesis of the amino acid threonine from aspartate, threonine itself acts as an inhibitor for the first enzyme in the pathway. When threonine levels are high, the pathway is shut down. When levels drop, the inhibitor detaches, and the pathway resumes.
Comparison of Inhibition Types
| Feature | Competitive Inhibition | Non-Competitive Inhibition |
|---|---|---|
| Inhibitor Binding Site | Active Site | Allosteric Site (site other than active site) |
| Inhibitor Structure | Structurally similar to substrate | Structurally different from substrate |
| Effect on Active Site | Physically blocks the active site | Changes the shape of the active site |
| Effect of Substrate Conc. | Can be overcome by increasing [Substrate] | Not affected by increasing [Substrate] |
| Reversibility | Usually reversible | Can be reversible or irreversible |
| Example | Malonate inhibiting succinate dehydrogenase | Cyanide, heavy metals, feedback inhibition |
Possible Questions and Answers
-
Q: What is the fundamental difference between competitive and non-competitive inhibition? A: The primary difference is the binding site. A competitive inhibitor binds to the active site, directly competing with the substrate. A non-competitive inhibitor binds to an allosteric site, changing the enzyme's shape indirectly.
-
Q: Why does increasing substrate concentration have no effect on non-competitive inhibition? A: Because the non-competitive inhibitor does not bind to the active site, it is not in competition with the substrate. Regardless of how many substrate molecules are present, the inhibitor can still bind to its allosteric site and change the enzyme's shape, rendering it inactive.
-
Q: What is feedback inhibition and why is it important for cells? A: Feedback inhibition is a cellular control mechanism where the end product of a metabolic pathway inhibits an enzyme at the beginning of that pathway. It is important because it prevents the overproduction of molecules, conserving energy and resources for the cell.
Biological Significance
Enzyme inhibition is essential for regulating metabolic pathways within cells. Medically, it is the principle behind many drugs (e.g., antibiotics like sulphonamides and penicillin) and the mechanism of action for many poisons.