1.7 Membrane Transport Mechanism
The main concepts related to the mechanisms controlling the movement of solutes (ions, small molecules) across a biological membrane. This transport is dependent on the membrane's permeability, solute concentration, and the size and charge of the solute.
Passive Transport
A type of transport that does not require cellular energy (ATP). Molecules move down a concentration gradient (from high to low concentration).
Simple Diffusion
- Definition: The movement of molecules from a region of higher concentration to a region of lower concentration, down a concentration gradient, without the aid of transport proteins.
- Concentration Gradient: The difference in the concentration of a substance between two regions.
- Mechanism: Small, non-polar molecules like oxygen (), carbon dioxide (), and water can pass directly through the phospholipid bilayer of the cell membrane.
- Function: Equalizes the concentration of these molecules inside and outside the cell. Each type of molecule moves down its own gradient independently.
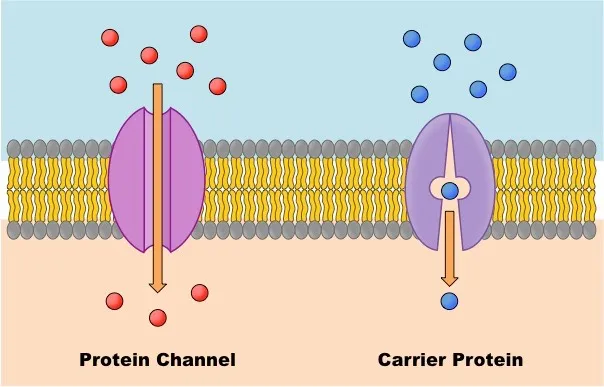
Facilitated Diffusion
- Definition: A type of passive transport in which molecules move from a region of higher concentration to lower concentration, assisted by transport proteins, without using ATP.
- Mechanism: Used for molecules that cannot easily cross the phospholipid bilayer — polar molecules, larger ions, and large molecules. It relies on two types of transport proteins:
- Channel Proteins: Create aqueous pores in the membrane for charged/polar molecules to pass through.
- Open Channel Proteins: Always open.
- Gated Channel Proteins: Can be opened or closed to regulate substance flow.
- Carrier Proteins: Bind to specific molecules, undergo a conformational (shape) change, and release the molecules on the other side of the membrane. Their rate is affected by temperature and saturation.
- Channel Proteins: Create aqueous pores in the membrane for charged/polar molecules to pass through.
Comparison: Simple vs. Facilitated Diffusion
| Feature | Simple Diffusion | Facilitated Diffusion |
|---|---|---|
| Type | Passive transport | Passive transport |
| Direction | Higher → Lower concentration | Higher → Lower concentration |
| Energy (ATP) | Not required | Not required |
| Transport proteins | Not required | Required (channel or carrier) |
| Rate | Generally slower | Faster, but limited by protein availability |
| Molecules transported | Small nonpolar (O₂, CO₂) | Polar molecules (glucose, amino acids), ions (Na⁺, Cl⁻) |
Osmosis
- Definition: The net movement of solvent molecules (typically water) from a solution of low solute concentration (high water potential) to a solution of high solute concentration (low water potential) through a semipermeable membrane.
- Semipermeable Membrane: A membrane that allows solvent molecules (like water) to pass through but not larger solute molecules (like sugar or salt).
- Related Concepts:
- Osmotic Pressure: The minimum pressure needed to prevent the inward flow of water across a semipermeable membrane.
- Water Potential: A measure of the tendency of water to move from one area to another. Pure water has the highest water potential.
- Tonicity Effects:
- Hypotonic solution (lower solute than cell): Water enters the cell → animal cell swells and may lyse; plant cell becomes turgid.
- Hypertonic solution (higher solute than cell): Water leaves the cell → animal cell shrinks (crenation); plant cell undergoes plasmolysis.
- Isotonic solution (equal solute): No net water movement.
Examples of Osmosis:
- Feeling thirsty after eating salty food.
- Swelling of raisins and seeds when soaked in water.
- Kidney dialysis.
Active Transport
- Definition: The movement of molecules or ions across a membrane from a region of lower concentration to a region of higher concentration, against a concentration gradient, requiring cellular energy.
- Mechanism: Requires energy in the form of ATP, carried out by specific membrane proteins (pumps).
Types of Active Transport
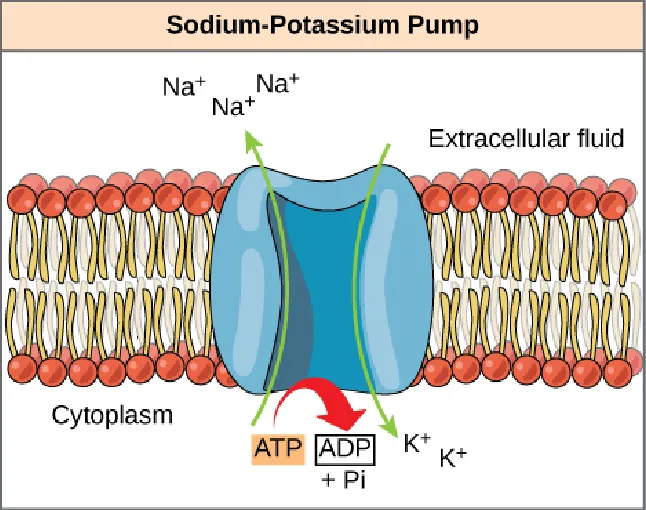
- Primary Active Transport:
- Directly uses chemical energy (ATP) to transport molecules against their gradient.
- Example: The Sodium-Potassium Pump (Na⁺/K⁺ pump) — pumps 3 Na⁺ ions out of the cell and 2 K⁺ ions in per cycle, against their respective gradients. This process consumes over one-third of a cell's total ATP.
- Secondary Active Transport:
- Uses an electrochemical gradient (created by primary active transport) as an indirect energy source.
- A transporter protein couples the movement of an ion (e.g., Na⁺) down its gradient to the movement of another molecule against its gradient.
Active Transport in Plants
- Root cells use active transport to absorb minerals and water from the soil, allowing the plant to accumulate nutrients at concentrations higher than in the surrounding soil.
Examples of Active Transport
- Movement of ions out of cardiac muscle cells.
- Transportation of amino acids across the intestinal lining.
- Secretion of proteins like enzymes and hormones.
- Mineral uptake by root hair cells in plants.
Summary Table
| Transport Type | Energy Required? | Direction | Proteins Required? | Examples |
|---|---|---|---|---|
| Simple Diffusion | No | High → Low | No | , |
| Facilitated Diffusion | No | High → Low | Yes (channel/carrier) | Glucose, Na⁺, Cl⁻ |
| Osmosis | No | High → Low water potential | No (aquaporins optional) | Water movement |
| Active Transport | Yes (ATP) | Low → High | Yes (pumps) | Na⁺/K⁺ pump, mineral uptake |
Biological Significance: Membrane transport is fundamental for life, enabling cells to acquire nutrients, eliminate waste products, maintain ion gradients, and communicate with their environment, thereby maintaining homeostasis.
<PracticeQuestions questionsString="[{"question":"What is the main difference between passive and active transport?","answer&q