2.7 Monosaccharides
Monosaccharides are the simplest form of carbohydrates, serving as fundamental energy sources and building blocks for more complex carbohydrates. They are defined as polyhydroxy aldehydes or polyhydroxy ketones.
Definition
A monosaccharide is a simple sugar that cannot be hydrolyzed into smaller carbohydrate units.
General Formula: , where is the number of carbon atoms, typically ranging from 3 to 7.
Structure: All carbon atoms except one have a hydroxyl group (-OH). The remaining carbon is part of either an aldehyde group (-CHO) or a ketone group (C=O).
Classification of Monosaccharides
Monosaccharides are classified based on two main criteria: the type of functional group they contain and the number of carbon atoms in their backbone.
Based on Functional Group
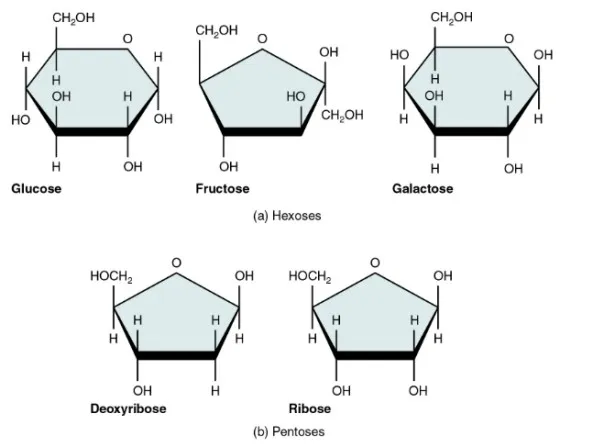
- Aldoses: Monosaccharides containing an aldehyde group. Example: Glucose, Ribose.
- Ketoses: Monosaccharides containing a ketone group. Example: Fructose, Ribulose.
Based on Number of Carbon Atoms
- Trioses (3C)
- Tetroses (4C)
- Pentoses (5C)
- Hexoses (6C)
- Heptoses (7C)
The following table combines both classification methods and highlights key examples and their biological functions.
| Class | Formula | Aldoses | Ketoses | Function |
|---|---|---|---|---|
| Trioses (3C) | Glyceraldehyde | Dihydroxyacetone | Intermediates in photosynthesis and cellular respiration. | |
| Tetroses (4C) | Erythrose | Erythrulose | Intermediates in bacterial photosynthesis. | |
| Pentoses (5C) | Ribose, Deoxyribose () | Ribulose | Ribose and deoxyribose are components of RNA and DNA respectively. Ribulose is an intermediate in photosynthesis. | |
| Hexoses (6C) | Glucose, Galactose | Fructose | Glucose is the primary respiratory fuel. Fructose is an intermediate in respiration. Galactose is a component of milk sugar (lactose). | |
| Heptoses (7C) | Glucoheptose | Sedoheptulose | Intermediates in photosynthesis. |
Properties of Monosaccharides
-
Cannot be Hydrolyzed: They are the simplest carbohydrate units.
-
Highly Soluble in Water: The multiple polar hydroxyl (-OH) groups form hydrogen bonds with water molecules.
-
Sweet Taste: Their specific structure activates sweet taste receptors on the tongue, which is why they are called "sugars".
-
Reducing Sugars: They possess a free aldehyde or ketone group that can donate electrons to reduce other compounds, while they themselves become oxidized.
-
Stereoisomerism: They can exist as different isomers due to the presence of asymmetric or chiral carbons (a carbon atom attached to four different groups), leading to different spatial arrangements of -H and -OH groups.
-
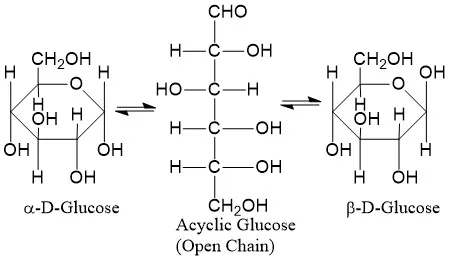
Exist in Two Structural Forms: Monosaccharides with 5 or more carbons can exist as an open-chain (acyclic) structure or a ring (cyclic) structure. The ring form is predominant in aqueous solutions.
Chemical Structures of Monosaccharides
In crystalline form, monosaccharides are typically open-chain structures. When dissolved in water, pentoses and hexoses spontaneously form stable ring structures.
Types of Ring Structures
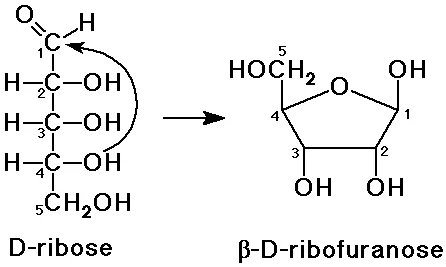
- Furanose: A five-membered ring containing four carbon atoms and one oxygen atom. This structure is formed by pentoses (e.g., ribose) and ketohexoses (e.g., fructose).
- Pyranose: A six-membered ring containing five carbon atoms and one oxygen atom. This structure is formed by aldohexoses (e.g., glucose).
Ring Formation
- In aldohexoses (like glucose), the aldehyde group on C1 reacts with the hydroxyl group on C5 to form a pyranose ring (glucopyranose).
- In aldopentoses (like ribose), the aldehyde group on C1 reacts with the hydroxyl group on C4 to form a furanose ring (ribofuranose).