2.6 Carbohydrates
Carbohydrates are organic compounds composed of carbon, hydrogen, and oxygen. The name literally means "hydrates of carbon," as they were initially thought to have a general formula of Cₙ(H₂O)ₙ. While this formula applies to some carbohydrates like glucose (), it is not a strict rule. For example, rhamnose () is a carbohydrate, while lactic acid () is not.
The modern chemical definition is:
"Organic compounds that are either polyhydroxy aldehydes or polyhydroxy ketones (monosaccharides) or their complex derivative compounds (polysaccharides) which upon hydrolysis produce either polyhydroxy aldehydes or polyhydroxy ketones or both."
Chemical Nature of Carbohydrates
Carbohydrates are structurally based on either an aldehyde group or a ketone group, with multiple hydroxyl (-OH) groups attached. This makes them polyhydroxy aldehydes or polyhydroxy ketones.
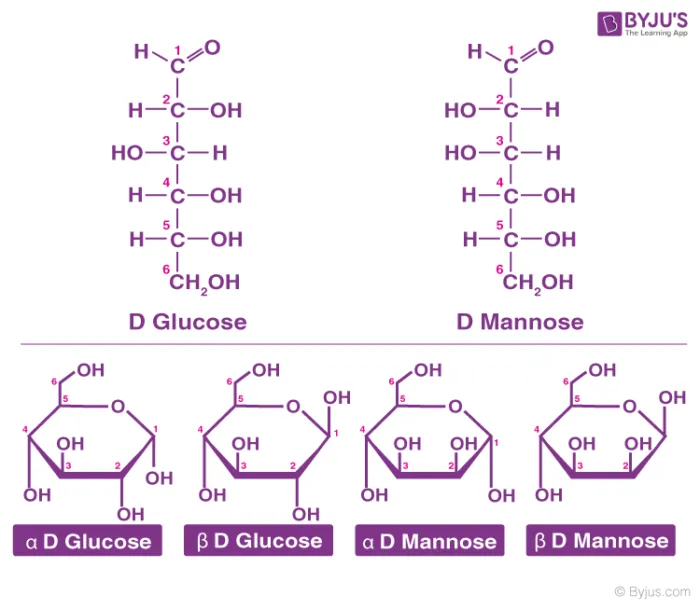
Classification of Carbohydrates
Carbohydrates are also known as sugars or saccharides (from the Greek word sakcharon, meaning sugar). Their classification is based on the number of saccharide units they contain.
They are generally classified into three main groups:
- Monosaccharides: Simple sugars
- Oligosaccharides: Short chains of sugars
- Polysaccharides: Complex carbohydrates
| Comparison of Carbohydrate Classes | Monosaccharides | Oligosaccharides | Polysaccharides |
|---|---|---|---|
| Saccharide Units | Consist of a single saccharide unit. | Composed of 2 to 10 saccharide units. | Composed of more than 10 (often hundreds or thousands) saccharide units. |
| Complexity and Hydrolysis | Simplest carbohydrates; cannot be hydrolyzed further. | Less complex; upon hydrolysis, they yield 2 to 10 monosaccharides. | Highly complex; upon hydrolysis, they yield many monosaccharides. |
| Solubility in Water | Highly soluble. | Less soluble. | Generally insoluble. |
| Taste | Sweetest among all carbohydrates. | Less sweet in taste. | Tasteless. |
Monosaccharides and Disaccharides
When two monosaccharides join, they form a disaccharide through a condensation reaction, resulting in a specific linkage.
Polysaccharides
Polysaccharides serve as energy storage or structural components. In plants, the Cell Wall→ is primarily composed of cellulose.
Possible Questions and Answers
Q: What is the modern chemical definition of a carbohydrate? A: An organic compound that is a polyhydroxy aldehyde or polyhydroxy ketone, or a complex compound that yields these upon hydrolysis.
Q: What is the basis for classifying carbohydrates? A: They are classified based on the number of saccharide (sugar) units they contain.
Q: How do the properties of solubility and taste change as carbohydrates become more complex? A: As the complexity increases from monosaccharides to polysaccharides, solubility in water and sweetness both decrease. Monosaccharides are highly soluble and sweet, while polysaccharides are generally insoluble and tasteless.