2.5 Water as a Medium of Life
Water is an essential constituent for all life on Earth, with its distribution being directly proportional to the abundance of life. Organisms are composed of approximately 70–90% water, with concentrations varying from 20% in seeds and bones to over 99% in jellyfish. Its unique properties make it the "medium of life," where most biochemical reactions occur.
Properties of Water
Water's vital role in biology is due to its unique chemical and physical properties, which stem from its polarity and the resulting hydrogen bonds.
1. High Polarity
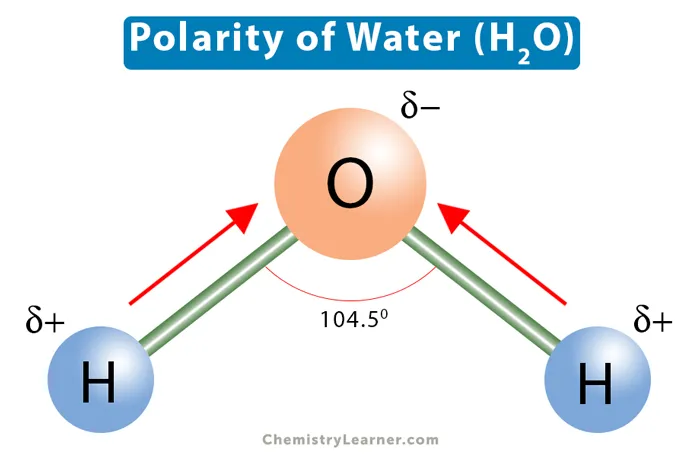
A water molecule () consists of one oxygen atom joined to two hydrogen atoms by polar covalent bonds. In these bonds, electrons are shared unequally. The more electronegative oxygen atom pulls the shared electrons closer, giving it a slight negative charge (), while the hydrogen atoms are left with slight positive charges (). This makes water a polar molecule.
- Universal Solvent: Because of its polarity, water is an excellent solvent for other polar and ionic substances (electrolytes). When dissolved, these substances dissociate into ions, which is a favourable state for chemical reactions.
- Biological Significance: This property ensures that nearly all biochemical reactions in cells occur in an aqueous medium.
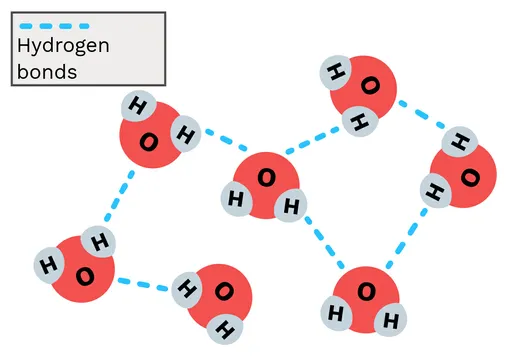
2. Hydrogen Bonding
The polarity of water molecules allows them to form weak attractions with each other. A hydrogen bond forms between the slightly positive hydrogen atom of one molecule and the slightly negative oxygen atom of a neighbouring molecule.
- State of Matter: These numerous but weak bonds are why water is a liquid at temperatures suitable for life.
- Biological Significance: Hydrogen bonding is responsible for many of water's other crucial properties, including cohesion, adhesion, and high specific heat capacity.
3. Cohesion and Adhesion
- Cohesion is the attraction between water molecules, causing them to stick together. This creates high surface tension.
- Adhesion is the attraction of water molecules to other polar surfaces.
- Biological Significance: Together, cohesion and adhesion allow water to be an effective transport medium, enabling it to move through the vascular tissues of plants (xylem) and the circulatory systems of animals.
4. High Specific Heat Capacity
Specific heat capacity is the amount of heat required to raise the temperature of 1 gram of a substance by 1°C. Water has a very high specific heat capacity (1 Cal/g/°C or 4.18 J/g/°C).
- Reason: A large amount of heat energy is first used to break the hydrogen bonds between water molecules before the temperature of the water itself can increase.
- Biological Significance: This property allows water to act as a temperature stabilizer (regulator). It protects organisms from sudden thermal changes, helping them maintain a stable internal temperature (homeostasis).
5. High Heat of Vaporization
Heat of vaporization is the amount of heat needed to convert a unit mass of a liquid into a gas. Water has a high heat of vaporization (574 calories/gram).
- Reason: Significant energy is required to break the hydrogen bonds and allow water molecules to escape as vapor.
- Biological Significance: This is a highly efficient cooling mechanism for organisms. When sweat or water evaporates from a surface (e.g., skin, leaves), it carries away a large amount of body heat, causing a cooling effect with minimal water loss. For example, the evaporation of just 2 mL of water can lower the temperature of 998 mL of remaining water by 1°C.
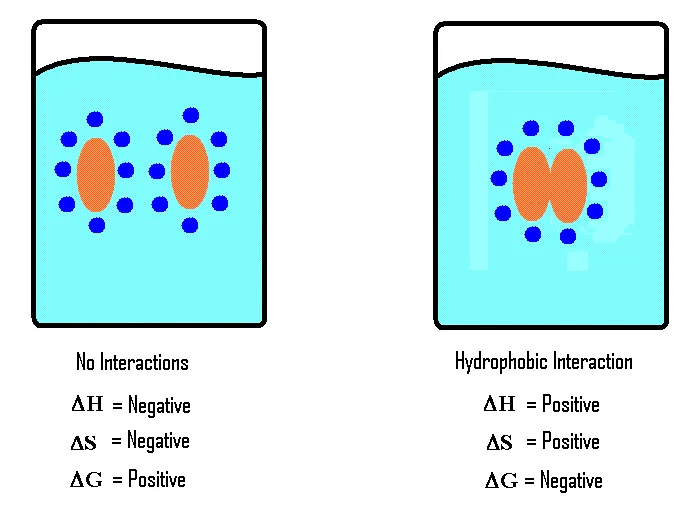
6. Hydrophobic Exclusion
Hydrophobic exclusion is the tendency of non-polar (hydrophobic) substances to aggregate and minimize their contact with water.
- Example: Oil droplets in water will merge to form a single large drop.
- Biological Significance: This principle is crucial for the formation and integrity of lipid bilayer membranes (cell membranes), as the hydrophobic tails of phospholipids spontaneously orient themselves away from the aqueous environment.
Cell Wall and Plasma Membrane→
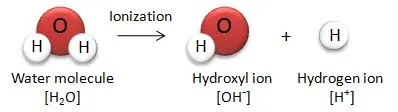
7. Ionization
Water molecules can reversibly dissociate (ionize) into a hydrogen ion () and a hydroxyl ion ():
- Biological Significance: These ions are highly reactive and participate in many cellular reactions. Their concentration determines the pH of a solution, which is critical for enzyme function and other biochemical processes.
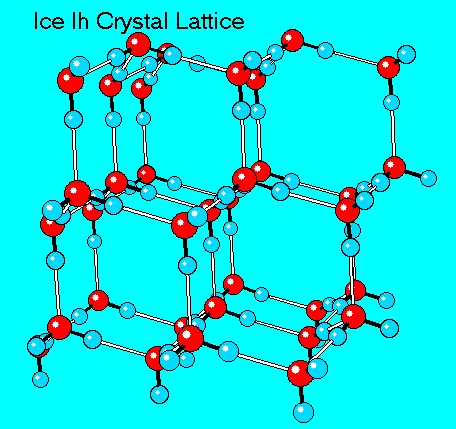
8. Lower Density of Ice
Unlike most substances, the solid form of water (ice) is less dense than its liquid form, which is why ice floats.
- Reason: As water freezes, its molecules form a crystal lattice structure. The hydrogen bonds hold the molecules a fixed distance apart, creating a more open, and therefore less dense, arrangement than in liquid water.
- Biological Significance: In freezing weather, a layer of ice forms on the surface of ponds and lakes. This ice acts as an insulating layer, protecting the aquatic life in the liquid water below from freezing.