2.8 Isomers and Stereoisomers of Glucose
A detailed breakdown of compounds with the same molecular formula but different structural arrangements, focusing on the various types of stereoisomers found in glucose.
Isomerism
A phenomenon where compounds share the same molecular formula but differ in their structural arrangement.
- Example: Glucose and Fructose both have the formula , but glucose is an aldo sugar (aldehyde group) while fructose is a keto sugar (ketone group). They are considered functional group isomers.
Stereoisomerism in Glucose
Stereoisomers are molecules with the same molecular formula and sequence of bonded atoms, but they differ in the three-dimensional orientation of their atoms in space. The number of possible stereoisomers can be calculated using the formula , where n is the number of asymmetric carbon atoms.
- For Glucose: Glucose has 4 asymmetric carbon atoms (C-2, C-3, C-4, C-5), so it has possible stereoisomers.
Key Carbon Atoms in Glucose Structure
- Asymmetric Carbon: A carbon atom that is bonded to four different atoms or groups. In glucose, C-2, C-3, C-4, and C-5 are asymmetric.
- Anomeric Carbon: The carbon atom in the ring structure that was formerly the carbonyl carbon (aldehyde or ketone group) in the open-chain form. In glucose, this is C-1.
- Penultimate Carbon: The second-to-last carbon in the chain, which is also the last asymmetric carbon. In glucose, this is C-5.
Types of Stereoisomers
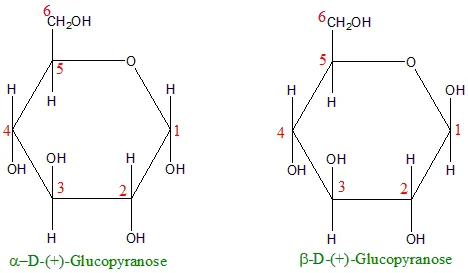
1. Anomers
- Stereoisomers that differ in the position of the -H and -OH groups only at the anomeric carbon (C-1).
- α-glucose: The -OH group on the anomeric carbon is pointing downward.
- β-glucose: The -OH group on the anomeric carbon is pointing upward.
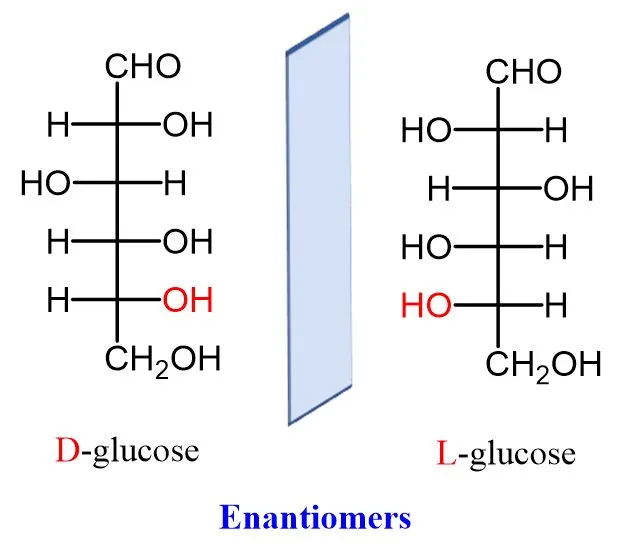
2. Enantiomers
- Stereoisomers that are non-superimposable mirror images of each other. The D and L forms of a sugar are enantiomers.
- D-glucose (Dextro): The "right-handed" form. The -OH group on the penultimate carbon (C-5) is on the right side.
- L-glucose (Levo): The "left-handed" form. The -OH group on the penultimate carbon (C-5) is on the left side.
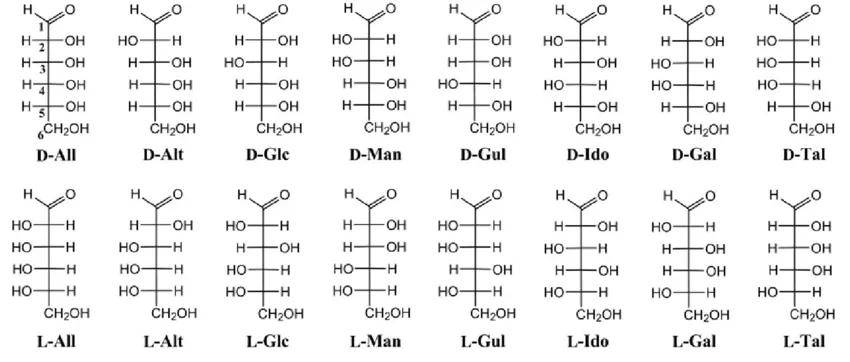
3. Diastereoisomers
- Stereoisomers that are not mirror images of each other. They differ in the arrangement of -H and -OH groups at more than one asymmetric carbon.
- Example: D-Glucose and D-Altrose are diastereoisomers.
4. Epimers
- A specific type of diastereoisomer. They differ in the arrangement of -H and -OH groups at only one asymmetric carbon (other than the anomeric carbon).
- Example: D-Glucose and D-Mannose are epimers, differing only at C-2. D-Glucose and D-Galactose are also epimers, differing only at C-4.
| Stereoisomer Type | Basis of Difference | Relationship | Example |
|---|---|---|---|
| Anomers | Position of -OH on anomeric carbon (C-1) | Isomers (α or β) | α-glucose vs. β-glucose |
| Enantiomers | Mirror images; position of -OH on penultimate carbon (C-5) | Non-superimposable mirror images (D or L) | D-glucose vs. L-glucose |
| Epimers | Position of -OH on a single asymmetric carbon | Not mirror images | D-glucose vs. D-Mannose (at C-2) |
| Diastereoisomers | Position of -OH on more than one asymmetric carbon | Not mirror images | D-glucose vs. D-Altrose |
Biological Significance
Living organisms almost exclusively use D-sugars. Enzymes, like those in the human digestive system, are stereospecific and can only metabolize D-sugars. This is why artificial sweeteners can be made from L-sugars, as the body cannot metabolize them for energy.