2.12 Proteins
Proteins are fundamental macromolecules that perform a vast array of critical roles in living organisms, essential for the structure, function, regulation, and maintenance of cells and body systems. They are the main structural components of the .
General Characteristics
- Proteins are the main structural components of the cell.
- All proteins are composed of Carbon (C), Hydrogen (H), Oxygen (O), and Nitrogen (N).
- Some proteins may also contain Phosphorus (P), Sulfur (S), Iron (Fe), Iodine (I), and Magnesium (Mg).
Structure of Proteins
Polymer Nature: Proteins are polymers, meaning they are large molecules made up of repeating smaller units.
Monomers: The building blocks (monomers) of proteins are amino acids.
Polypeptide Chains: A protein is chemically a chain of amino acids linked together, known as a polypeptide chain. A functional protein can consist of one or more polypeptide chains.
Amino Acids
Definition: Amino acids are the fundamental building blocks of all proteins. While many exist, only 20 are commonly found in biological proteins.
General Structure: Each amino acid has a central carbon atom, called the alpha (α) carbon, which is bonded to four different groups:
- A hydrogen atom (-H)
- An amino group (-NH₂)
- A carboxyl group (-COOH)
- A variable R-group (or side chain)
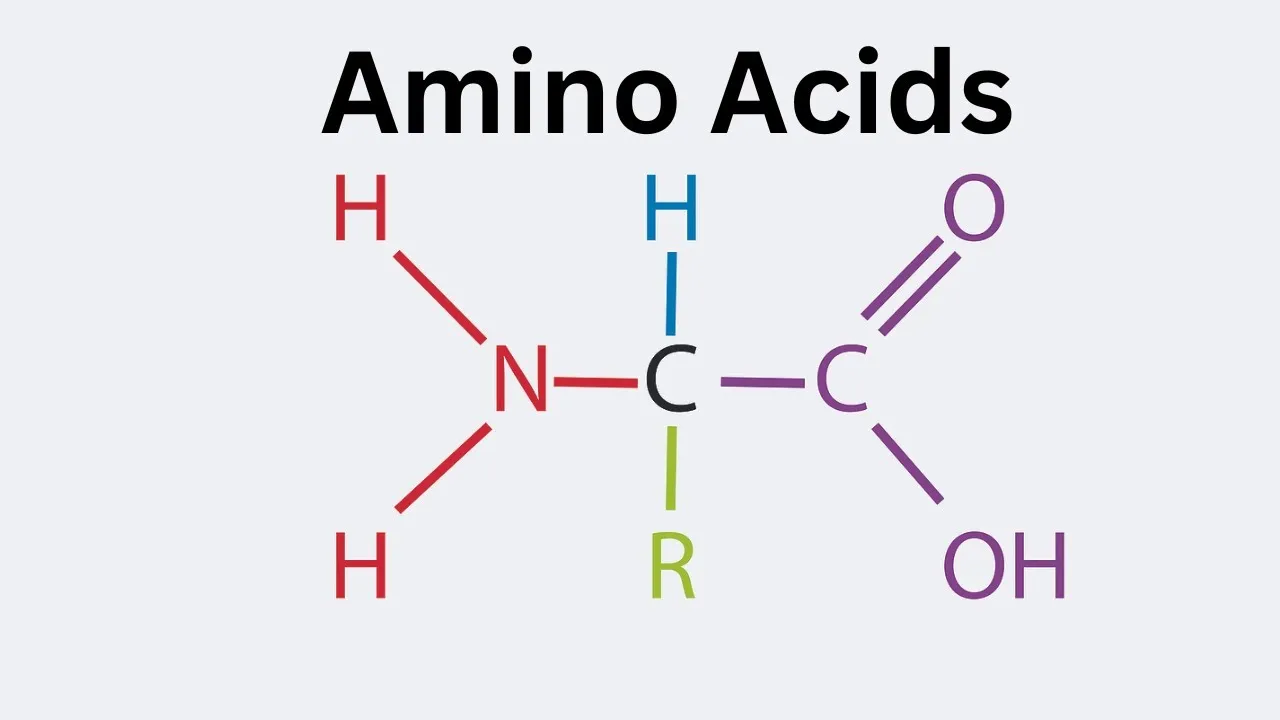
The R-Group: The 'R' group is unique to each of the 20 amino acids and determines its specific chemical properties (e.g., whether it is acidic, basic, polar, or nonpolar).
Essential and Non-essential Amino Acids
Amino acids are categorized based on whether the human body can synthesize them.
| Category | Definition | Source | Examples | Functions |
|---|---|---|---|---|
| Essential Amino Acids | Amino acids that cannot be synthesized by the human body and must be obtained through diet. | Milk, eggs, fish, meat, vegetables, lentils. | Lysine, leucine, isoleucine, histidine, methionine, phenylalanine, threonine, valine, tryptophan (9 total). | Build and repair muscle/bone; act as precursors for hormones and neurotransmitters. |
| Non-essential Amino Acids | Amino acids that can be synthesized by the human body, typically by modifying other molecules. | Synthesized in the body. | Alanine, arginine, asparagine, aspartic acid, cysteine, glutamic acid, glutamine, glycine, proline, serine, tyrosine (11 total). | Detoxification; synthesis of red and white blood cells (RBCs & WBCs); support brain and cardiac function. |
Levels of Protein Organization
Proteins exhibit complex three-dimensional structures categorized into four levels:
Primary Structure
The linear sequence of amino acids in a polypeptide chain. Even a single change in this sequence can lead to disorders like Sickle Cell Anemia.
Secondary Structure
The folding or coiling of the polypeptide chain into specific shapes like the -helix or -pleated sheet, primarily maintained by hydrogen bonds.
Tertiary and Quaternary Structure
Tertiary structure refers to the overall 3D shape of a single polypeptide, while Quaternary structure involves the arrangement of multiple polypeptide chains (e.g., Hemoglobin).
Classification of Proteins
Proteins can be classified based on their shape and solubility:
- Fibrous Proteins: Long, strand-like, water-insoluble proteins (e.g., Keratin, Collagen).
- Globular Proteins: Spherical, water-soluble proteins (e.g., Enzymes, Antibodies, Hemoglobin).
Denaturation
Protein denaturation is the process where a protein loses its native shape and function due to external stress like heat or pH changes.