2.13 Synthesis and Breakage of Peptide Linkages
This section outlines the formation and breakdown of peptide bonds, the fundamental linkages in proteins, and explores the resulting levels of protein structural organization.
Peptide Bond
A peptide bond is the covalent bond formed between two amino acids. The formation and breakage of these bonds are crucial for creating and breaking down proteins.
Synthesis of Peptide Bonds
- Process: Condensation (or dehydration synthesis).
- Location: Occurs on ribosomes during a process called translation, where genetic instructions from mRNA are used to build a protein.
- Mechanism:
- The hydroxyl (-OH) group from the carboxyl group of one amino acid is removed.
- A hydrogen (-H) atom from the amino group of another amino acid is removed.
- These combine to form a water molecule ().
- A covalent bond forms between the carbon atom of the first amino acid's carboxyl group and the nitrogen atom of the second amino acid's amino group.
- Products:
- Dipeptide: A molecule formed from two linked amino acids.
- Polypeptide: A long chain of amino acids linked by many peptide bonds.
Structure of a Dipeptide
A dipeptide has two distinct ends:
- N-terminal end: The end with a free amino group ().
- C-terminal end: The end with a free carboxyl group (). New amino acids are typically added to this end to lengthen the chain.
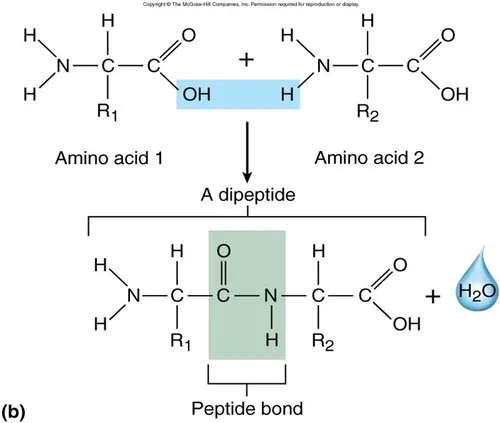
Breakage of Peptide Bonds
- Process: Hydrolysis (the addition of water to break a bond).
- Biological Context:
- Digestion: In the gut, enzymes like pepsin and trypsin break down dietary proteins into amino acids for absorption.
- Cellular Catabolism: Within cells, lysosomal enzymes can break down proteins to recycle amino acids for new protein synthesis or for energy (ATP) production.
Structural Conformations in Proteins
Once synthesized, a polypeptide chain folds into a specific three-dimensional shape, which is essential for its function. There are four levels of protein structural organization. For more on how these proteins are utilized in the cell, see Ultrastructure of Animal and Plant Cells→.
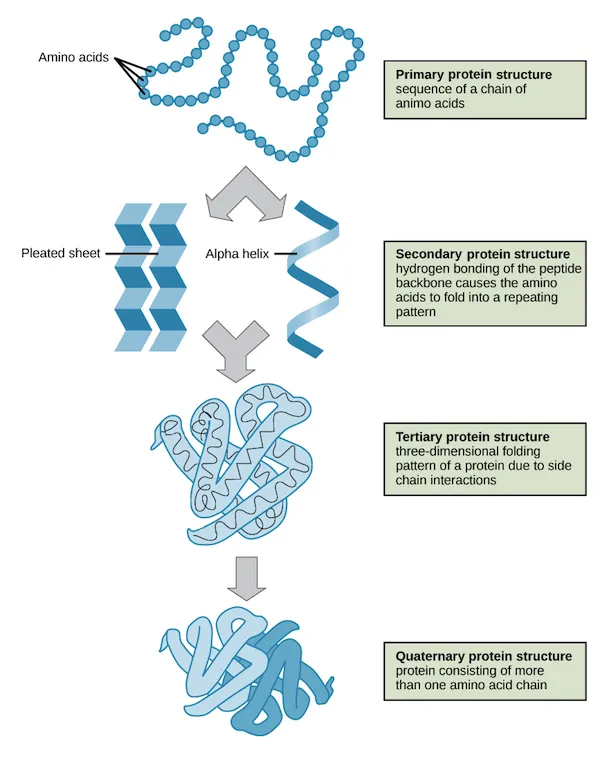
1. Primary Structure
- Description: The linear sequence and specific number of amino acids in the polypeptide chain.
- Bonding: Held together by peptide bonds.
- Significance: This sequence is determined by the genetic code (DNA) and dictates all higher levels of structure.
2. Secondary Structure
- Description: The initial folding or coiling of the primary polypeptide chain.
- Common Forms:
- α-helix: A spiral or helical coil.
- β-pleated sheet: A flattened, folded sheet-like structure.
- Bonding: Stabilized by hydrogen bonds between the backbone atoms of different amino acids (not the R-groups).
3. Tertiary Structure
- Description: The complex, three-dimensional folding of the secondary structure into a specific globular shape.
- Bonding: Stabilized by various interactions between the R-groups of the amino acids, including ionic bonds, disulfide bridges, hydrogen bonds, and hydrophobic interactions.
- Significance: This 3D shape is crucial for the protein's specific biological function.
4. Quaternary Structure
- Description: The arrangement and combination of two or more polypeptide chains (subunits) to form a single, functional protein complex.
- Bonding: Held together by interactions between the different subunits, such as hydrophobic interactions.
- Example: Hemoglobin, which consists of four polypeptide subunits.
| Structure | Description | Key Bonds Involved |
|---|---|---|
| Primary | Linear sequence of amino acids | Peptide bonds |
| Secondary | Coiling (α-helix) or folding (β-sheet) of the chain | Hydrogen bonds (between backbone) |
| Tertiary | 3D folding of the polypeptide into a globular shape | R-group interactions (ionic, disulfide, etc.) |
| Quaternary | Association of multiple polypeptide subunits | Hydrophobic interactions, etc. (between subunits) |