2.11 Polysaccharides
Polysaccharides are complex carbohydrates (polymers) that yield more than ten monosaccharide units upon hydrolysis. They are generally insoluble in water, not sweet, and play crucial roles in energy storage and structural support.
Key Concepts
Classification of Polysaccharides
- Homopolysaccharides: Composed of only one kind of monosaccharide monomer.
- Examples: Starch, glycogen, cellulose, chitin.
- Heteropolysaccharides: Composed of different kinds of monosaccharide monomers.
- Examples: Agar, pectin, peptidoglycan.
[FC: fc_cg8fpi]
[FC: fc_25seie]
[MCQ: f87d46e8-9043-46e0-8c55-f69b5595e492]
[MCQ: c47278d3-035f-4c6a-98c3-d8032ee235bd]
General Functions and Properties
Polysaccharides are ideal storage molecules for several reasons:
- Insolubility: Their large size makes them mostly insoluble in water, preventing them from exerting osmotic or chemical influence within the cell.
- Compactness: They can fold into compact shapes, allowing for efficient storage.
- Hydrolysis: They can be easily hydrolyzed (broken down) into simple sugars when energy is required.
- Primary Roles:
- Food and Energy Storage: Starch (in plants) and Glycogen (in animals).
- Structural Material: Cellulose (in plants) and Chitin (in fungi and arthropods).
[FC: fc_1isszv]
Specific Polysaccharides
1. Starch
- Definition: A homopolysaccharide made from the condensation of hundreds of α-glucose monomers.
- Function: The primary energy storage carbohydrate in plants.
- Storage Locations: Found mainly in roots, stems, and seeds (e.g., cereal grains, potato tubers).
- Digestion: Broken down by the enzyme amylase into maltose, which is then broken down by maltase into glucose.
- Identification Test: Gives a blue-black color with iodine solution.
- Structure: Starch consists of two types of molecules:
- Amylose: An unbranched, linear chain of glucose units linked by α-1,4-glycosidic bonds. It is soluble only in hot water.
- Amylopectin: A branched structure. It has a main linear chain with α-1,4-glycosidic bonds and branches attached by α-1,6-glycosidic bonds. It is completely insoluble in water.
[FC: fc_oksl03]
[FC: fc_htjqs8]
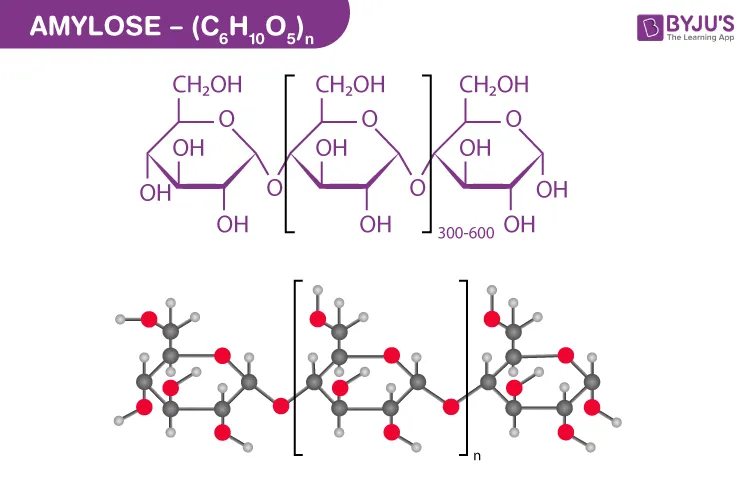 Figure 2.21: Structure of starch (Amylose)
Figure 2.21: Structure of starch (Amylose)
2. Glycogen
- Definition: A homopolysaccharide composed of multiple α-glucose monomers, often called "animal starch".
- Function: The primary energy storage carbohydrate in animals.
- Storage Locations: Stored mainly in the liver and muscles.
- Structure: Similar in structure to amylopectin but is much more branched.
- Identification Test: Gives a red color with iodine solution.
[MCQ: de67a5d5-d1b3-4f36-a51b-ec1216f08e44]
[PP: w5751cz5hq8b7cs93msg0enbvh82z4wa]
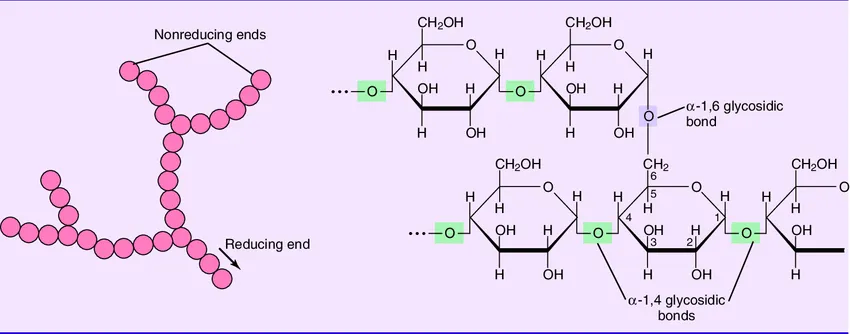 Figure 2.22: Structure of glycogen
Figure 2.22: Structure of glycogen
3. Cellulose
- Definition: The most abundant carbohydrate on Earth, a homopolysaccharide formed from the condensation of hundreds of β-glucose monomers.
- Function: A key structural component of plant cell walls.
- Examples: Cotton and paper are pure forms of cellulose.
- Structure: An unbranched, linear chain of glucose units linked by β-1,4-glycosidic bonds.
- Digestion: Humans cannot digest cellulose but it acts as dietary fiber (roughage). Herbivores can digest it with the help of symbiotic bacteria that produce the enzyme cellulase.
- Identification Test: Shows no color change with iodine solution.
[MCQ: 65fd9720-81e1-41f2-ab45-92d1832acabe]
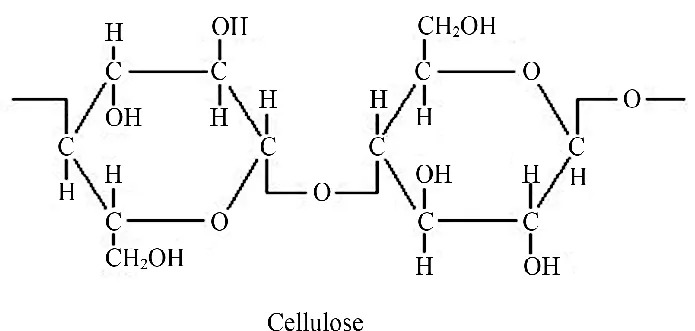 Figure 2.23: Structure of cellulose
Figure 2.23: Structure of cellulose
4. Chitin
- Definition: The second most abundant organic molecule, a homopolysaccharide also known as "fungal cellulose".
- Function: A structural carbohydrate found in the cell walls of fungi and the exoskeletons of arthropods.
- Monomer: A modified form of glucose called N-acetyl glucosamine.
- Structure: An unbranched, linear chain of monomers linked by β-1,4-glycosidic bonds.
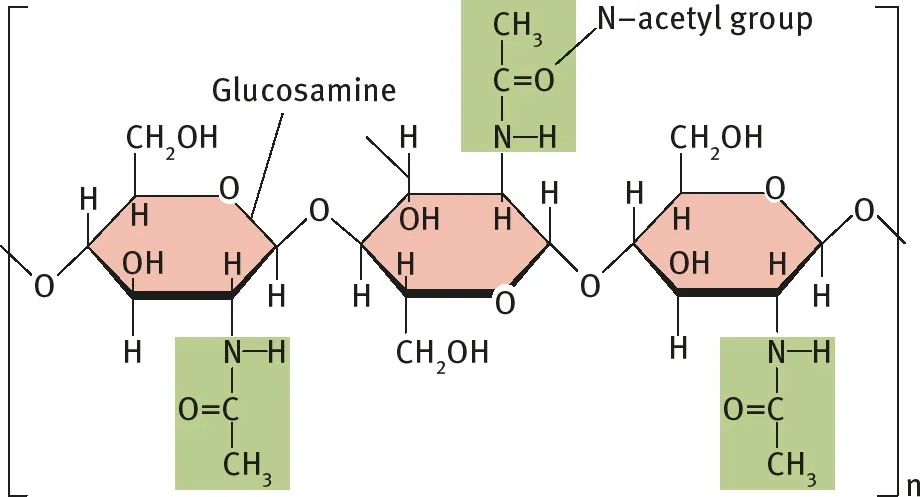 Figure 2.24: Structure of chitin
Figure 2.24: Structure of chitin
Comparison of Major Polysaccharides
| Feature | Starch | Glycogen | Cellulose | Chitin |
|---|---|---|---|---|
| Monomer | α-glucose | α-glucose | β-glucose | N-acetyl glucosamine |
| Bond Type | α-1,4 and α-1,6 | α-1,4 and α-1,6 | β-1,4 | β-1,4 |
| Structure | Branched (amylopectin) and Unbranched (amylose) | Highly Branched | Unbranched (linear) | Unbranched (linear) |
| Organism | Plants | Animals, Fungi | Plants | Fungi, Arthropods |
| Function | Energy Storage | Energy Storage | Structural | Structural |
| Iodine Test | Blue-black color | Red color | No change | No change |
Biochemical Tests
| Test | Procedure | Positive Result | Inference |
|---|---|---|---|
| Iodine Test for Starch | Add a few drops of iodine solution to the sample. | Dark blue-black coloration. | Starch is present. |
| Iodine Test for Glycogen | Add a few drops of iodine solution to the sample. | Red coloration. | Glycogen is present. |
| Benedict's Test for Glucose | Add Benedict's solution to the sample and boil. | Dirty green, yellow, or red precipitate. | Glucose (a reducing sugar) is present. |
| Fehling's Test for Glucose | Add Fehling's solution to the sample and heat. | Red precipitate. | Glucose (a reducing sugar) is present. |
| Molisch's Test for Sugars | Add alcoholic alpha-naphthol, then slowly add conc. . | Purple ring at the junction. | Carbohydrate (e.g., disaccharide) is present. |
Possible Questions and Answers
-
Q: What is the main difference between a homopolysaccharide and a heteropolysaccharide?
- A: A homopolysaccharide is made of a single type of monosaccharide monomer (e.g., starch from glucose), while a heteropolysaccharide is made of different types of monosaccharide monomers.
-
Q: Why are starch and glycogen excellent molecules for energy storage?
- A: They are large, insoluble molecules that do not affect the cell's osmotic balance. They are also compact and can be easily broken down (hydrolyzed) into glucose when energy is needed.
-
Q: Why cannot humans digest cellulose, and why is it still important in our diet?
- A: Humans lack the enzyme cellulase needed to break the β-1,4-glycosidic bonds in cellulose. It is important in the diet as dietary fiber or roughage, which aids in digestion and prevents abnormal food absorption.
-
Q: How does the structure of amylopectin differ from glycogen?
- A: Both are branched polymers of α-glucose. However, glycogen is much more highly branched than amylopectin.
Summary
- Polysaccharides are large polymers of monosaccharides used for energy storage and structural support.
- Homopolysaccharides (e.g., starch, glycogen, cellulose) consist of a single type of monomer.
- Starch (α-glucose; α-1,4 and α-1,6 bonds) is the energy store in plants.
- Glycogen (α-glucose; α-1,4 and α-1,6 bonds) is the highly branched energy store in animals.
- Cellulose (β-glucose; β-1,4 bonds) is an unbranched structural component of plant cell walls.
- Chitin (N-acetyl glucosamine; β-1,4 bonds) is an unbranched structural component of fungal cell walls and arthropod exoskeletons.
- The specific monomer and the type of glycosidic linkage (α or β) determine the polymer's overall structure and function.
The structural differences between polysaccharides dictate their biological roles, from providing a readily available source of energy to forming the robust, rigid structures essential for life.
References
(Derived from FBISE textbook)