2.10 Glycosidic Bonds in Disaccharides
This section details the formation, naming, and types of covalent bonds that link monosaccharide units together to form larger carbohydrates like disaccharides and polysaccharides.
Glycoside
A ring-structured sugar molecule (hexose or pentose) that is typically attached to another sugar molecule.
Glycosidic Bond
A covalent chemical bond formed between two monosaccharide units through a condensation reaction. This bond is crucial for the formation of disaccharides, oligosaccharides, and polysaccharides.
- Formation: A hydroxyl (-OH) group from one sugar reacts with a hydroxyl (-OH) group of another sugar. This process releases one molecule of water (), forming a C-O-C bridge between the two sugars.
- Naming Convention: Glycosidic bonds are named based on two factors:
- The carbon numbers of the two interacting carbons (e.g., C-1 and C-4).
- The anomeric form (alpha or beta) of the first sugar involved in the linkage.
- Example: An α-1,4 glycosidic linkage forms when the first sugar is an alpha anomer and its C-1 links to the C-4 of the second sugar.
Reducing vs. Non-Reducing Sugars
- Reducing Sugar: A carbohydrate that can be oxidized and cause the reduction of other substances. They have a free anomeric carbon (open chemical structure).
- Examples: All monosaccharides, maltose, lactose.
- Non-Reducing Sugar: A carbohydrate that cannot be oxidized. Both anomeric carbons are involved in the glycosidic bond, so no free anomeric carbon is available.
- Examples: Sucrose, all polysaccharides.
Types of Glycosidic Linkages in Disaccharides
The structure and properties of a disaccharide are determined by its constituent monosaccharides and the specific glycosidic bond that links them.
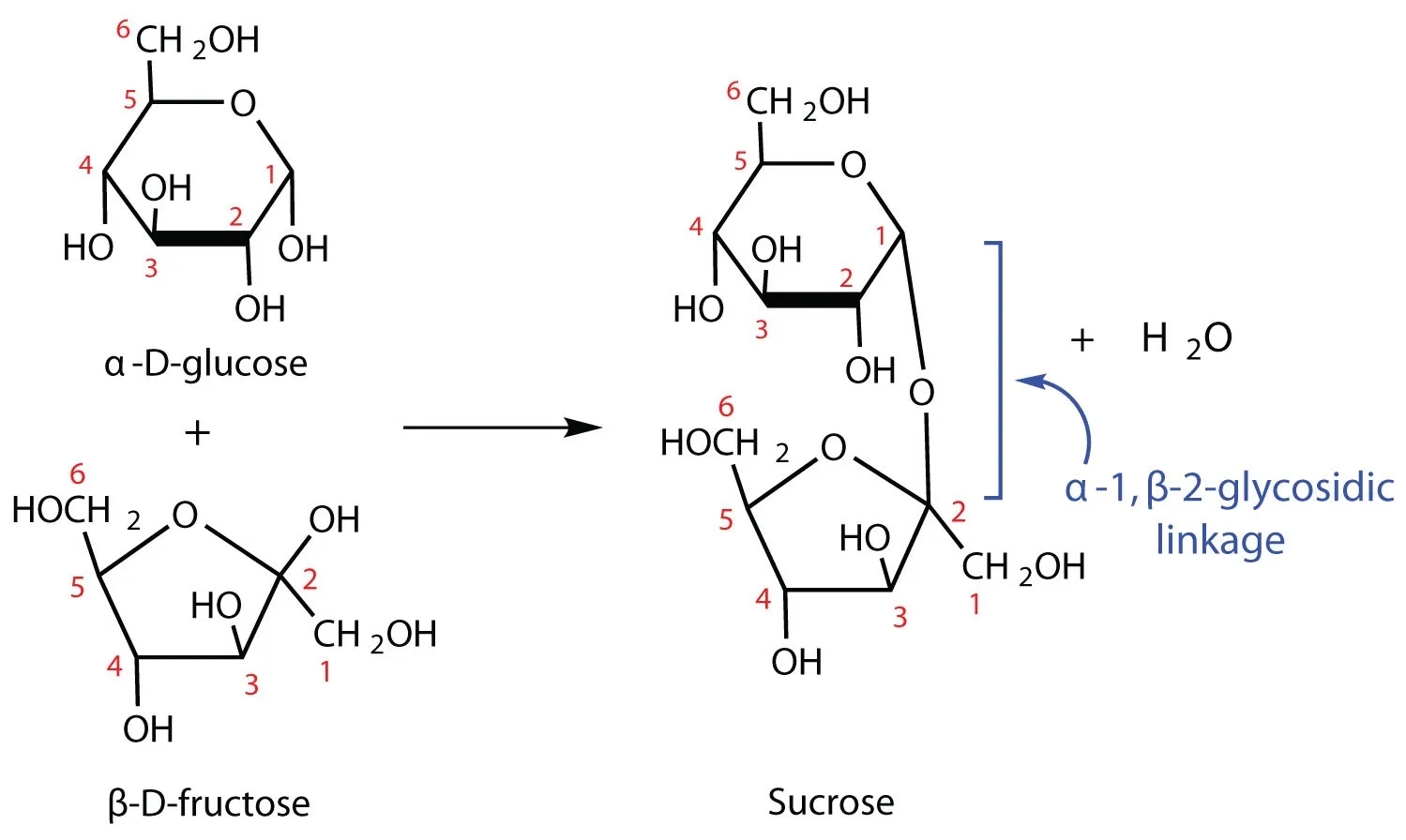
1. Glycosidic Linkage in Sucrose
- Monomers: Formed by the condensation of α-glucose and β-fructose.
- Reaction: The -OH group at C-1 of α-glucose reacts with the -OH group at C-2 of β-fructose.
- Bond Type: α-1,2-glycosidic linkage (also written as α-1, β-2-glycosidic linkage).
- Reducing status: Non-reducing sugar (both anomeric carbons are involved in the bond).
- Diagram:
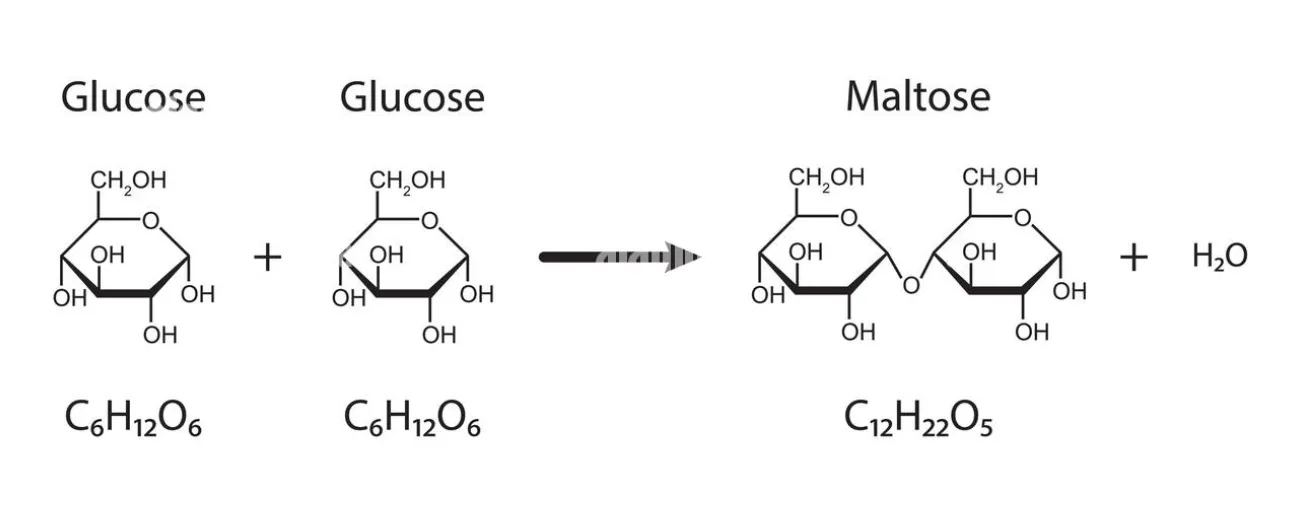
2. Glycosidic Linkage in Maltose
- Monomers: Formed by the condensation of two α-glucose molecules.
- Reaction: The -OH group at C-1 of the first α-glucose reacts with the -OH group at C-4 of the second α-glucose.
- Bond Type: α-1,4-glycosidic linkage.
- Reducing status: Reducing sugar (one anomeric carbon remains free).
- Diagram:
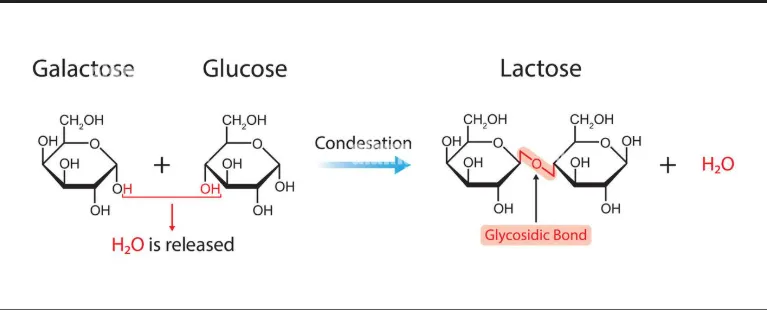
3. Glycosidic Linkage in Lactose
- Monomers: Formed by the condensation of β-galactose and β-glucose.
- Reaction: The -OH group at C-1 of β-galactose reacts with the -OH group at C-4 of β-glucose.
- Bond Type: β-1,4-glycosidic linkage.
- Reducing status: Reducing sugar (the anomeric carbon of glucose remains free).
- Diagram:
Summary Table
| Disaccharide | Monomer 1 | Monomer 2 | Glycosidic Bond Type | Reducing/Non-Reducing |
|---|---|---|---|---|
| Sucrose | α-glucose | β-fructose | α-1,2 | Non-reducing |
| Maltose | α-glucose | α-glucose | α-1,4 | Reducing |
| Lactose | β-galactose | β-glucose | β-1,4 | Reducing |
Biological Significance: The formation of glycosidic bonds allows for the synthesis of complex carbohydrates essential for energy storage (e.g., starch, glycogen) and structural support (e.g., cellulose, chitin).