8.13 Thermal Equilibrium and Internal Energy
Thermal Equilibrium
Thermal equilibrium is a fundamental concept in thermodynamics that describes the state of a system in which there is no net flow of thermal energy. When two or more objects are in thermal contact, heat flows naturally from the hotter object to the colder object. This process continues until they reach the same temperature, at which point they are said to be in thermal equilibrium.
Key Concept: Two systems are in thermal equilibrium if and only if they are at the same temperature.
- Diathermic Substances: Materials that allow heat to pass through them are called diathermic. Thermal equilibrium is established when objects are connected by a diathermic substance.
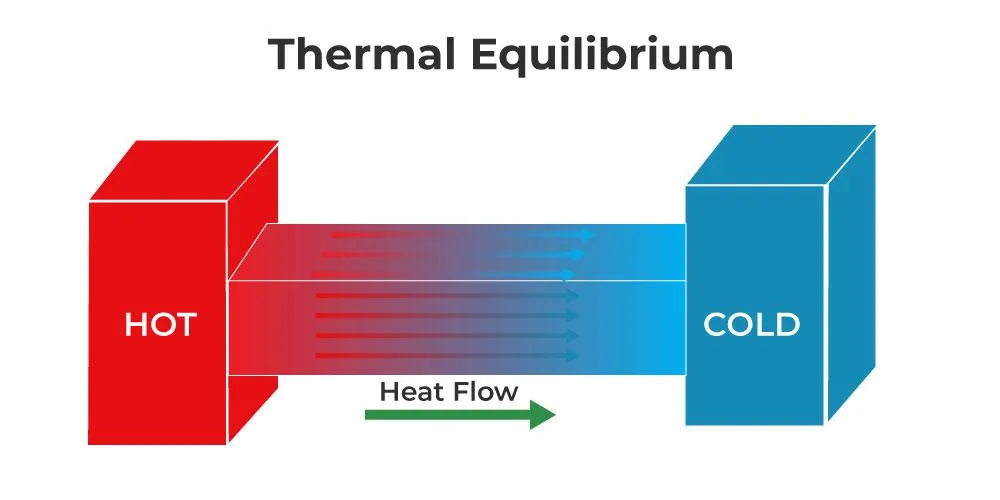
Internal Energy (U)
Internal energy is the total energy contained within a thermodynamic system. It is the sum of all the microscopic kinetic and potential energies of the particles (atoms and molecules) that make up the system.
- Kinetic Energy: This includes the translational (straight-line motion), rotational, and vibrational energy of the molecules.
- Potential Energy: This is the energy associated with the intermolecular forces (bonds) between the particles.
Internal Energy of an Ideal Gas
For an ideal gas, the intermolecular forces are assumed to be negligible. Therefore, the internal energy of an ideal gas consists almost entirely of the translational kinetic energy of its molecules.
- Kinetic Theory of Gases: This theory states that the average kinetic energy of the gas molecules is directly proportional to the absolute temperature () of the gas.
Where:
-
is the Boltzmann constant ( J/K).
-
Internal Energy and Temperature: Because the internal energy of an ideal gas is the sum of the kinetic energies of its molecules, the internal energy of an ideal gas depends only on its temperature.
Changing Internal Energy
The internal energy of a system can be changed in two ways:
- Heat Transfer (): Adding heat to a system increases its internal energy.
- Work Done (): Doing work on a system (e.g., compressing a gas) increases its internal energy.
Key Point: Internal energy is a state function, meaning its value depends only on the current state of the system (e.g., its temperature and pressure), not on how it got to that state.
This concept is further developed in the 1st Law of Thermodynamics→.
Sign Conventions for Internal Energy ()
- If the temperature of a system increases, its internal energy increases ().
- If the temperature decreases, its internal energy decreases ().
- If the temperature remains constant (an isothermal process), the change in internal energy for an ideal gas is zero ().
The Ideal Gas Law
The Ideal Gas Law is an equation of state that describes the relationship between the pressure (), volume (), temperature (), and the amount of an ideal gas ().
Equation:
Where:
- = Absolute pressure of the gas
- = Volume of the gas
- = Number of moles of the gas
- = The universal gas constant (8.314 J/mol·K)
- = Absolute temperature of the gas (in Kelvin)
An ideal gas is a theoretical gas that perfectly follows this law. Real gases approximate this behavior at low pressures and high temperatures.
The Ideal Gas Law with Boltzmann's Constant
The law can also be written in terms of the total number of molecules () in the gas and the Boltzmann constant ().
The Boltzmann constant () is a fundamental constant that relates the average kinetic energy of particles in a gas with the temperature of the gas. It connects the macroscopic world (described by ) and the microscopic world (described by , Avogadro's number).
Possible Questions and Answers
Q: What is the difference between heat and internal energy?
A: Internal energy is the total energy contained within a system. Heat is the energy that is transferred between a system and its surroundings due to a temperature difference. Heat is energy in transit, not a property that a system "has."
Q: When a substance melts, its temperature stays constant, yet heat is being added. Where does this energy go?
A: The added energy is used to increase the potential energy of the molecules by breaking the bonds that hold them in a fixed solid structure. Since the kinetic energy of the molecules does not increase (as temperature is constant), the internal energy of the substance still increases, but it is due to a change in potential energy, not kinetic energy.