8.12 The Gas Laws
The gas laws are a set of fundamental principles that describe the relationship between the macroscopic properties of a gas: pressure (), volume (), and temperature (), for a given amount of gas (). These laws were developed through experimental observations and form the basis of our understanding of gas behavior. They are combined in the Ideal Gas Law.
Pressure is a Derived Unit→ measured in Pascals (Pa).
Boyle's Law (Constant Temperature)
Boyle's Law describes the relationship between pressure and volume when the temperature and the amount of gas are held constant.
Statement: For a fixed mass of gas at constant temperature, the volume is inversely proportional to the pressure.
This means that if you increase the pressure on a gas, its volume will decrease proportionally, and vice versa.
Mathematical Formulation: For a gas changing from an initial state (1) to a final state (2):
Graphical Representation: The graph of pressure versus volume for an isothermal (constant temperature) process is a hyperbola. The graph of vs is a straight line through the origin.
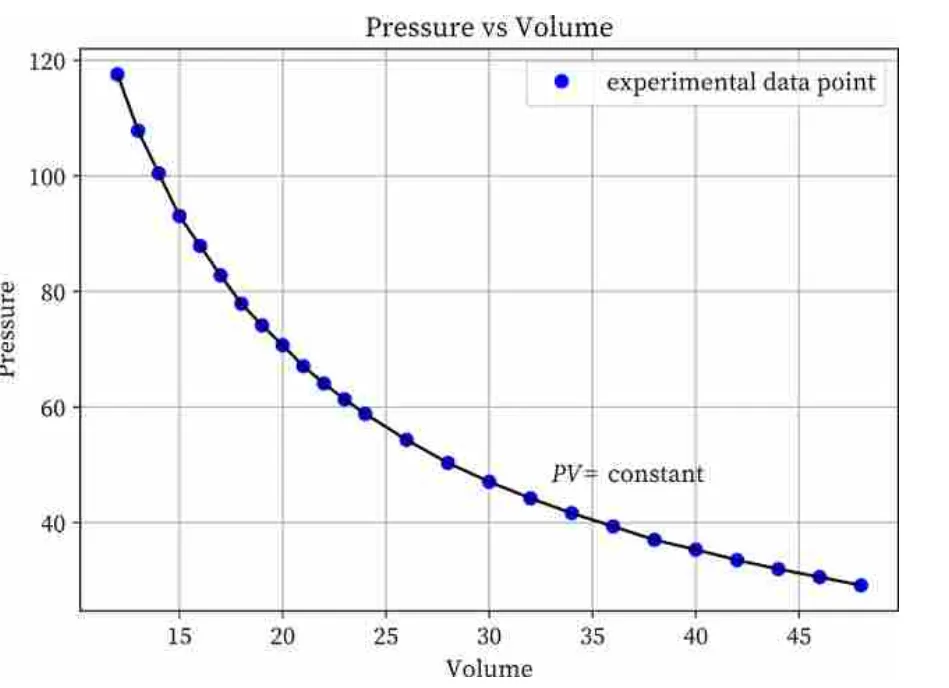
Charles's Law (Constant Pressure)
Charles's Law describes the relationship between volume and temperature when the pressure and the amount of gas are held constant.
Statement: For a fixed mass of gas at constant pressure, the volume is directly proportional to its absolute temperature (in Kelvin).
This means that heating a gas will cause it to expand, and cooling it will cause it to contract.
Mathematical Formulation: For a gas changing from an initial state (1) to a final state (2):
Important: Temperature must always be in Kelvin (K) for gas law calculations. Convert using .
Graphical Representation: The graph of volume versus absolute temperature is a straight line passing through the origin, confirming the direct proportionality .
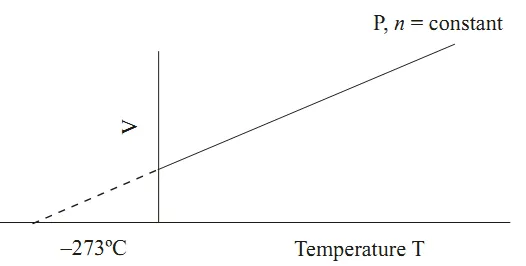
Gay-Lussac's Law (Constant Volume)
Gay-Lussac's Law (also known as Amontons's Law) describes the relationship between pressure and temperature when the volume and the amount of gas are held constant.
Statement: For a fixed mass of gas at constant volume, the pressure is directly proportional to its absolute temperature (in Kelvin).
This explains why the pressure inside a sealed container of gas increases when it is heated.
Mathematical Formulation: For a gas changing from an initial state (1) to a final state (2):
Ideal Gas Equation
By combining Boyle's Law, Charles's Law, and Avogadro's Law, we derive the General Gas Equation (Equation of State for an Ideal Gas):
Where:
- = pressure (Pa)
- = volume (m³)
- = number of moles (mol)
- = Universal Gas Constant
- = absolute temperature (K)
An equivalent form using the number of molecules and the Boltzmann constant :
For an ideal gas, the internal energy depends solely on temperature.
Worked Example: A gas occupies at a pressure of and temperature . Find the number of moles.
Summary Table
| Law | Constant Variable | Relationship | Formula |
|---|---|---|---|
| Boyle's Law | Temperature | ||
| Charles's Law | Pressure | ||
| Gay-Lussac's Law | Volume |