8.11 Thermodynamic Processes: Reversible, Irreversible, and Cyclic
In thermodynamics, a process describes the path a system takes as it transitions from one equilibrium state to another, typically involving an exchange of energy. Understanding the nature of these processes - whether they are idealized and can be reversed, or real-world and unidirectional - is fundamental to applying the laws of thermodynamics.
1. Reversible Process
A reversible process is an idealized process that can be reversed to return both the system and its surroundings to their exact original states. It is a theoretical concept used as a benchmark for analyzing thermodynamic efficiency.
Conditions for Reversibility:
- The process must occur infinitely slowly (quasi-statically), so the system is always in thermal and mechanical equilibrium.
- There must be no dissipative forces, such as friction, viscosity, or electrical resistance, that would convert useful energy into heat.
Characteristics:
- It is a theoretical ideal that cannot be perfectly achieved in practice.
- It represents the most efficient possible path for a process.
- The net change in the entropy of the universe (system + surroundings) is zero.
Examples:
- The extremely slow, frictionless compression or expansion of a gas.
- The gradual melting of ice into water, or freezing of water into ice, where the temperature difference is infinitesimally small.
2. Irreversible Process
An irreversible process is any process that is not reversible. Once it has occurred, it is impossible to return both the system and its surroundings to their original states. All real-world, spontaneous processes are irreversible.
Causes of Irreversibility:
- Dissipative Forces: Friction, viscosity, and other forms of energy dissipation convert work into heat that cannot be fully recovered.
- Sudden Changes: Processes that happen quickly (for example, a rapid expansion of gas into a vacuum, an explosion) are inherently irreversible.
- Heat Transfer across a Finite Temperature Difference: Heat flowing from a hot object to a cold one is a spontaneous, irreversible process.
Characteristics:
- All natural processes are irreversible.
- They are less efficient than their corresponding reversible counterparts.
- The total entropy of the universe always increases during an irreversible process.
Examples:
- Burning a piece of wood.
- Mixing sugar into coffee.
- Heat flowing from a hot stove to a cold pot (conduction).
- Any process involving friction.
| Feature | Reversible Process | Irreversible Process |
|---|---|---|
| Nature | Idealized, theoretical | Real-world, natural |
| Speed | Infinitely slow | Occurs at a finite speed |
| Dissipation | No friction or other dissipative forces | Dissipative forces are present |
| Entropy | Total entropy of the universe is constant | Total entropy of the universe increases |
| Path | Can be exactly retraced in reverse | Cannot be exactly retraced |
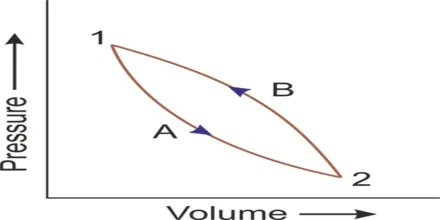
3. Cyclic Process
A cyclic process is a series of thermodynamic processes that returns a system to its initial state. After completing the cycle, all of the system's state properties (pressure, volume, temperature, internal energy) are the same as they were at the beginning.
Key Property: Since the initial and final states are identical, the net change in internal energy () over one complete cycle is always zero.
First Law Application: Applying the 1st Law of Thermodynamics→ () to a cyclic process:
This means the net heat absorbed by the system in a cycle is equal to the net work done by the system during that cycle. Work and heat are measured in Joules.
Relevance: Cyclic processes are the basis for all Heat Engines and Refrigerators, which are designed to operate continuously.
Possible Questions and Answers
Q: Why are reversible processes important if they don't actually happen in reality?
A: Reversible processes serve as an essential theoretical tool. They define the maximum possible efficiency for any thermodynamic process, such as in a heat engine (the Carnot Cycle). By comparing a real, irreversible process to its ideal, reversible counterpart, engineers can measure and improve the efficiency of real-world devices.
Q: For a heat engine operating in a cycle, where does the work output come from if the internal energy of the working substance doesn't change?
A: In a cyclic process, the net work output comes from the conversion of heat energy. The engine absorbs a certain amount of heat from a high-temperature source, converts part of it into work, and rejects the rest to a low-temperature sink. The net heat absorbed () is equal to the net work done.