8.1 First Law of Thermodynamics
The First Law of Thermodynamics expresses the law of conservation of energy. Energy cannot be created or destroyed; it can only be transferred or transformed from one form to another. In a thermodynamic system, the law relates the change in internal energy to the heat added to the system and the work done by the system.
Heat and Internal Energy
When heat () is supplied to a system, it can produce two effects: it can increase the system's internal energy (), which is the sum of all microscopic kinetic and potential energies of its particles, and it can cause the system to perform work () on its surroundings.
For an ideal gas, internal energy depends only on temperature. Therefore, internal energy is directly proportional to temperature.
Equation of the First Law
The relationship is expressed mathematically as:
Where:
- is the net heat added to the system.
- is the change in the system's internal energy.
- is the net work done by the system.
Sign Conventions:
- is positive when heat is added to the system.
- is positive when work is done by the system (expansion).
- is positive when the internal energy increases.
Examples of the First Law
- Melting Ice: Heat energy is transferred from the surroundings to an ice cube, increasing its internal energy to break the bonds of its solid structure and change its state to liquid water.
- Sweating: Your body transfers heat to sweat. This heat provides the energy for the sweat to evaporate, thus removing heat from your body and lowering your temperature.
- Human Metabolism: The chemical energy in food is converted through metabolic processes. This energy is used to do work and to maintain the body's internal energy, with the excess released as heat.
See Applications Of 1st Law Of Thermodynamics→ for further applications of this law.
Thermodynamic Processes
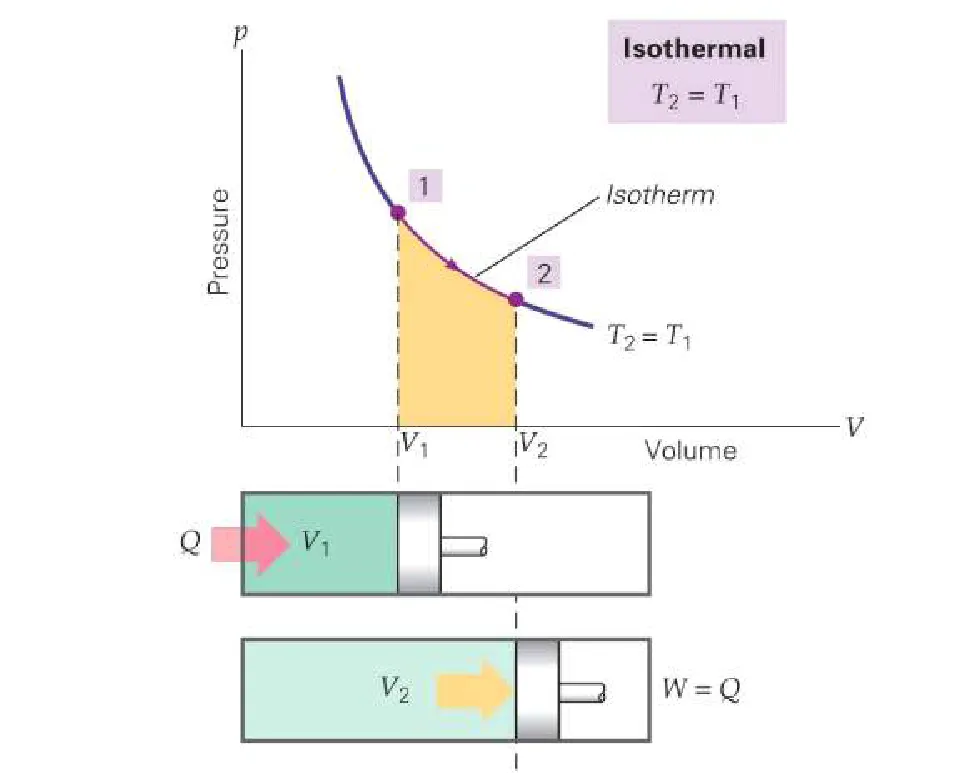
1. Isothermal Process
An isothermal process occurs at constant temperature. For an ideal gas, internal energy depends only on temperature, so if temperature is constant, the change in internal energy is zero.
- Condition:
- First Law Equation: Since , the first law simplifies to:
Any heat added to the system is entirely converted into work done by the system.
- Governing Law: For a gas, this process follows Boyle's Law ().
- Example: The very slow expansion or compression of a gas in a container in thermal contact with a large heat reservoir, or the melting of ice at 0°C.
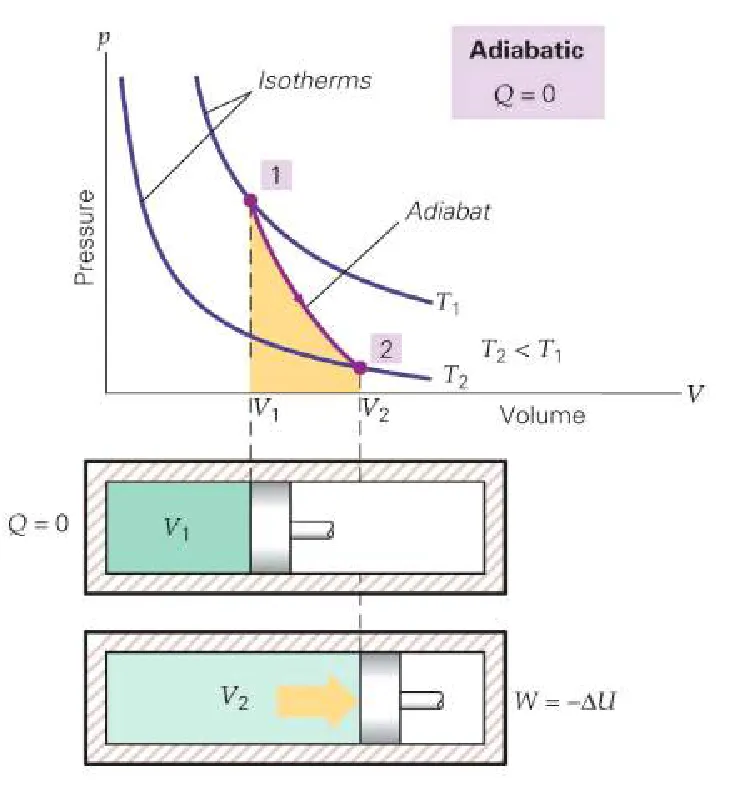
2. Adiabatic Process
An adiabatic process is one where no heat is transferred into or out of the system. This occurs when the system is perfectly insulated or when the process happens very rapidly.
- Condition:
- First Law Equation: Since , the first law becomes:
Any work done by the system comes at the expense of its own internal energy, typically causing its temperature to change.
- Example: The rapid compression of air in a bicycle pump or the expansion of gas in the cylinder of an engine.
See Adiabatic Equation→ for the equation governing this process.
3. Isochoric Process
An isochoric process occurs at constant volume. Since volume does not change, the system does no work on its surroundings.
- Condition: , which means
- First Law Equation: Since , the first law simplifies to:
All heat added to the system goes into increasing its internal energy.
- Example: Heating a gas in a sealed, rigid container.
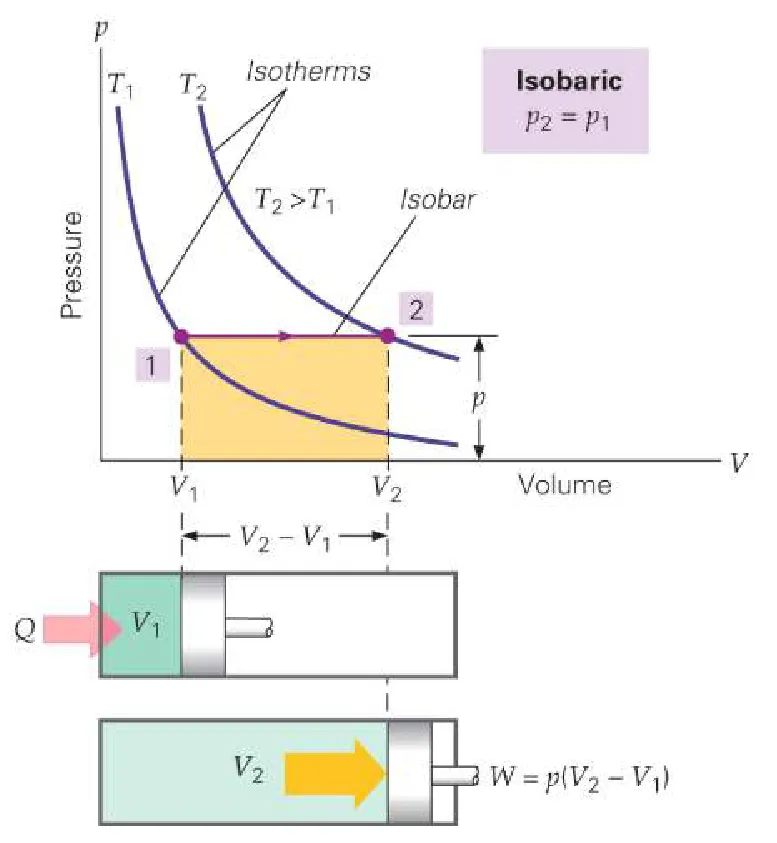
4. Isobaric Process
An isobaric process occurs at constant pressure. In this process, heat can be exchanged, work can be done, and internal energy can change.
- Condition: Pressure () is constant.
- Work Done: The work done by the system during an expansion is given by:
-
First Law Equation: The full form applies: .
-
Example: Boiling water in an open container where pressure remains constant at atmospheric pressure.
| Process | Key Characteristic | First Law Expression | Example |
|---|---|---|---|
| Isothermal | Constant temperature | Melting of ice at 0°C | |
| Adiabatic | No heat transfer | Compression of air in a piston | |
| Isochoric | Constant volume | Heating gas in a sealed container | |
| Isobaric | Constant pressure | Heating water in an open pot |
The 2nd Law Of Thermodynamics→ builds upon the first law by introducing the concept of entropy and the direction of natural processes.