11.3 Sulphur
Sulphur is a non-metal element located below oxygen in Group 16 (VIA) of the periodic table. At room temperature, elemental sulphur is a bright yellow, crystalline solid. Sulphur atoms typically form stable, cyclic octatomic molecules with the chemical formula .
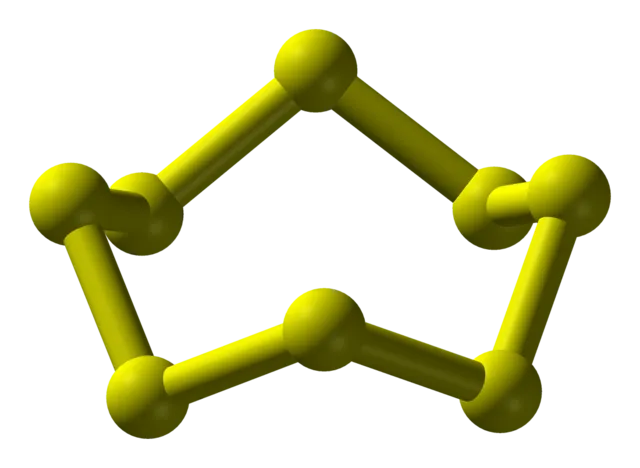
Electronic Configuration and Stability
Sulphur has 6 electrons in its valence shell. According to the Rules of Electronic Configuration→, its valence shell configuration is . This leads to possible oxidation states of -2, +2, +4, and +6.
- Most Stable Oxidation State: The +6 oxidation state is the most stable for sulphur. This is due to its ability to expand its octet using vacant 3d-orbitals, allowing it to form six bonds (e.g., in or ).
- Examples: Sulphur in the +6 state is found in important compounds like sulphuric acid () and sulphate ions ().
- Molecular Stability: The elemental molecule is relatively inert because the single S-S bonds are strong and require significant energy to break.
11.3.1 Allotropes of Sulphur
Sulphur exists in several allotropic forms. The two main crystalline allotropes are:
| Allotrope | Also Called | Stable Temperature Range | Structure |
|---|---|---|---|
| Rhombic sulphur | Alpha () sulphur | Below | puckered rings, orthorhombic crystal |
| Monoclinic sulphur | Beta () sulphur | Above (up to m.p.) | puckered rings, monoclinic crystal |
The temperature is the transition temperature at which the two forms are in equilibrium. Rhombic sulphur is the thermodynamically stable form at room temperature.
11.3.2 Properties of Sulphur
Sulphur is a versatile element that can function as both an oxidising agent and a reducing agent, depending on the chemical reaction.
- As an Oxidising Agent: Sulphur can gain two electrons to achieve a stable octet (like a noble gas), forming the sulphide ion (). Its oxidation state decreases from 0 to -2.
- As a Reducing Agent: Sulphur can also lose electrons to exist in higher oxidation states like +4 and +6. Its oxidation state increases from 0 to +4 or +6.
11.3.3 Sulphur as an Oxidising Agent
In these reactions, sulphur's oxidation state is reduced from 0 to -2.
1. Reaction with Hydrogen
Sulphur combines directly with hydrogen gas upon heating to form hydrogen sulphide.
2. Reaction with Metals
Sulphur reacts with active metals like zinc and iron on heating to form metal sulphides.
In these reactions, the oxidation state of sulphur decreases from 0 to -2, while the oxidation state of the metals (Zn and Fe) increases from 0 to +2.
11.3.4 Sulphur as a Reducing Agent
In these reactions, sulphur's oxidation state increases from 0 to +4 or +6.
1. Reaction with Oxygen (Combustion)
Sulphur burns in air with a characteristic blue flame to produce sulphur dioxide gas.
In the presence of moist air, sulphur can be slowly oxidised to sulphuric acid.
2. Reaction with Oxidising Agents
When treated with strong oxidising agents like concentrated nitric acid () or concentrated sulphuric acid (), sulphur is oxidised.
-
Reaction with Nitric Acid: Here, sulphur is oxidised from 0 to +6, while nitrogen in nitric acid is reduced from +5 to +4.
-
Reaction with Sulphuric Acid: In this reaction, elemental sulphur is oxidised from 0 to +4, while the sulphur in sulphuric acid is reduced from +6 to +4.
11.3.5 Uses of Sulphur and its Compounds
Sulphur and its compounds have a wide range of important industrial and chemical applications.
- Fertiliser Production: Sulphuric acid and ammonium sulphate are crucial components in fertilisers, providing essential sulphur for the synthesis of amino acids and vitamins in plants.
- Gunpowder: Gunpowder is a mixture of sulphur, charcoal, and potassium nitrate (). Sulphur acts as a fuel, lowers the ignition temperature, and increases the combustion rate.
- Rubber Industry: Sulphur is used in the vulcanization of rubber, a process that cross-links polymer chains to improve the rubber's strength, elasticity, and durability.
- Synthetic Organic Chemistry: Sulphur-containing functional groups are vital in organic synthesis. For example, thiols (-SH) and sulfonyl chlorides are used to create pharmaceuticals, including a class of antibiotics known as sulfonamides.
- Dyes and Pigments: Sulphur compounds are used in the manufacturing of various dyes and pigments.
- Food Preservatives: Sulphur dioxide () and sulphites (e.g., ) act as reducing agents that inhibit oxidation, preventing spoilage and preserving the color of foods and beverages like wine and dried fruits.