11.4 Sulphuric Acid
Sulphuric acid () is a colourless, oily liquid. It is a dense substance with a density of , making it almost twice as heavy as water. It solidifies at into colourless crystals. It is often referred to as the "King of Chemicals" due to its vast industrial applications.
11.4.1 Contact Process (Manufacture of Sulphuric Acid)
The industrial production of sulphuric acid is carried out using the Contact Process. This process relies on a few key raw materials and a series of controlled chemical reactions.
Raw Materials: Sulphur (S), Air (providing Oxygen, ), and Water (). Sulphur extracted from fossil fuel impurities is also a common source.
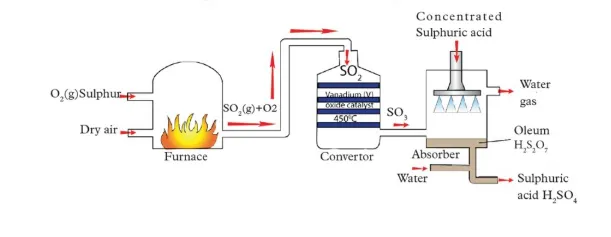
Steps of the Contact Process
1. Oxidation/Burning of Sulphur
Sulphur is burned in an excess of air to produce sulphur dioxide gas.
2. Purification of Sulphur Dioxide
The sulphur dioxide gas must be purified to remove impurities like arsenic oxide. These impurities can "poison" the catalyst used in the next step, rendering it ineffective.
3. Catalytic Oxidation of Sulphur Dioxide
The purified sulphur dioxide is mixed with more air (oxygen) and passed over a catalyst to form sulphur trioxide. This is a reversible and exothermic reaction.
Optimal Conditions: According to Le Chatelier's Principle, the following conditions favor the maximum yield of :
- Temperature:
- Pressure: 2–3 atmospheres (atm)
- Catalyst: Vanadium(V) oxide ()
Note: Sulphur trioxide () is not dissolved directly in water. The reaction is extremely exothermic and would release a large amount of heat, causing the mixture to boil violently and create a hazardous acid mist.
4. Production of Oleum and Dilution
To manage the reaction safely, sulphur trioxide is first dissolved in concentrated sulphuric acid to form a substance called oleum (or fuming sulphuric acid).
The oleum is then carefully diluted with water in a controlled manner to produce concentrated sulphuric acid of the desired concentration.
11.4.2 Properties of Sulphuric Acid
Sulphuric acid exhibits several key chemical properties, making it a versatile industrial chemical.
1. Ionization Reaction
In an aqueous solution, sulphuric acid ionizes to produce hydrogen ions and sulphate ions.
2. Reaction with Bases (Neutralization)
As a strong acid, it reacts with bases to form a salt and water.
3. Reaction with Active Metals
Dilute sulphuric acid reacts with active metals (e.g., Zinc) to produce a metal salt and hydrogen gas.
4. Reaction with Metal Carbonates
It reacts with metal carbonates to form a salt, water, and carbon dioxide gas.
5. As a Dehydrating Agent
Concentrated sulphuric acid has a strong affinity for water and can remove it from other compounds, often charring organic matter.
-
Decomposition of formic acid:
-
Dehydration of ethanol to form ethene:
6. As an Oxidizing Agent
Hot, concentrated sulphuric acid is a powerful oxidizing agent.
- Reaction with Metals: It oxidizes metals like copper, liberating sulphur dioxide gas.
In this reaction, copper is oxidized (oxidation state from to ), and sulphur in is reduced (oxidation state from to in ).
- Reaction with other compounds:
11.4.3 Uses of Sulphuric Acid
Sulphuric acid is one of the most important and widely produced industrial chemicals due to its versatility. Its major applications include:
- Production of fertilizers (e.g., ammonium sulphate)
- Manufacturing detergents, paints, pigments, and dyes
- Production of plastics and synthetic fibers (e.g., rayon)
- As an electrolyte in lead-acid car batteries
- Tanning of leather
- Cleaning metal surfaces ("pickling") before electroplating or galvanizing