6.5 Born-Haber Cycle
The Born-Haber cycle is a specific application of Hess's Law used to determine the lattice energies of binary ionic compounds (like ).
Lattice Energy
The lattice energy () is the change in energy that occurs when one mole of an ionic solid is formed from its separated gaseous ions. It is a measure of the strength of the ionic bonds in a crystal lattice.
Lattice energy cannot be measured directly in an experiment. However, it can be calculated indirectly using the Born-Haber cycle, which breaks down the formation of an ionic compound into a series of well-defined steps.
Constructing the Born-Haber Cycle for NaCl
We can illustrate the cycle using sodium chloride () as an example. The standard enthalpy of formation () for is . The cycle connects this value to other measurable enthalpy changes.
The overall formation reaction is:
This process can be broken down into five distinct steps:
Step I: Sublimation of Sodium Solid sodium is converted into gaseous sodium atoms.
Step II: Ionization of Sodium Gaseous sodium atoms lose an electron to form gaseous sodium ions. This is the first ionization energy () of sodium. Ionization Energy→
Step III: Dissociation (Atomization) of Chlorine The covalent bond in chlorine molecules is broken to form gaseous chlorine atoms. We only need one mole of Cl atoms, so we start with half a mole of .
Step IV: Formation of Chloride Ion Gaseous chlorine atoms gain an electron to form gaseous chloride ions. This is the electron affinity () of chlorine.
Step V: Formation of the Ionic Lattice Gaseous sodium and chloride ions combine to form one mole of solid sodium chloride. This is the lattice energy () that we want to calculate.
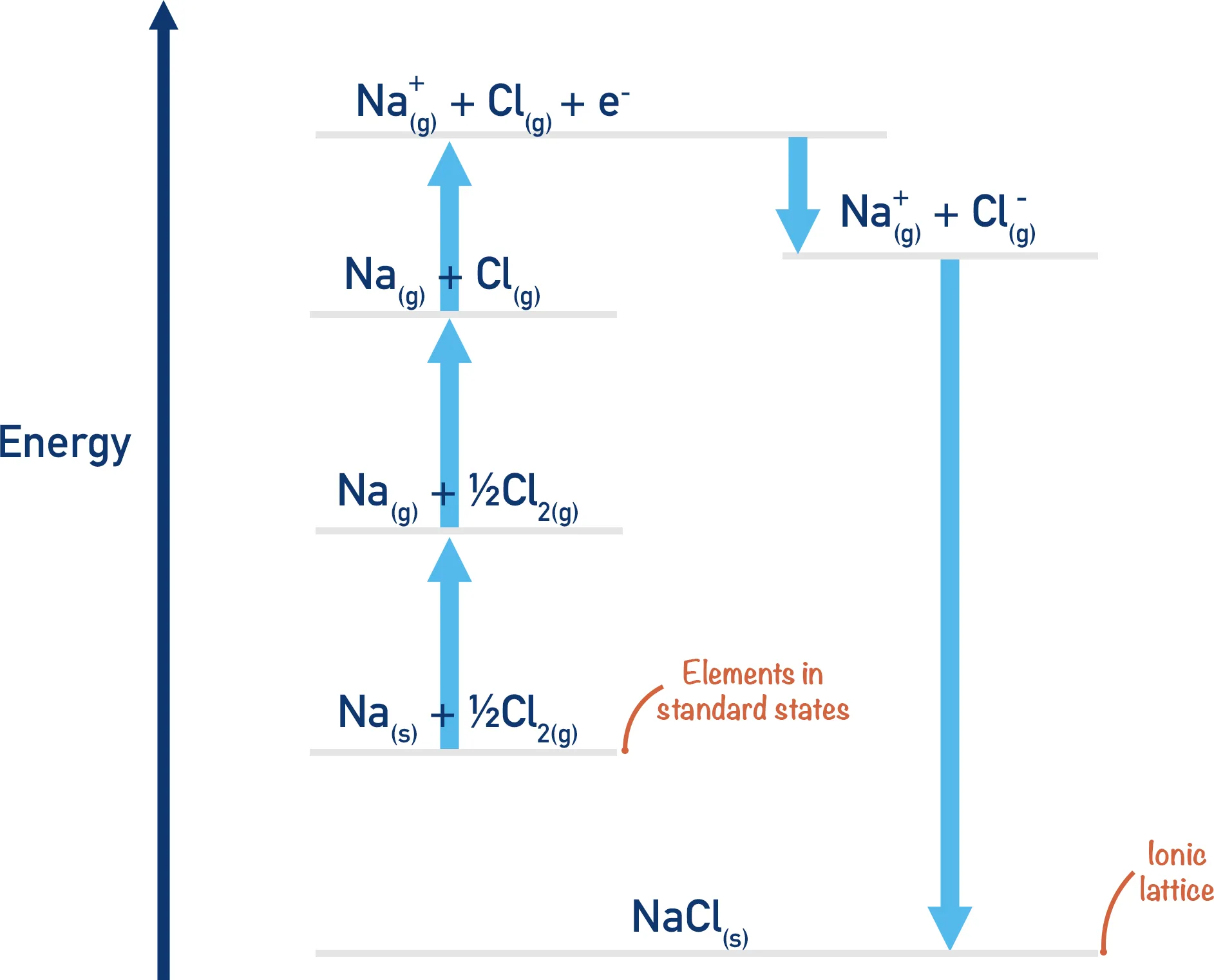
Calculation of Lattice Energy for NaCl
According to Hess's Law, the total enthalpy change of the direct route (enthalpy of formation) must equal the sum of the enthalpy changes of the indirect route (the five steps).
-
Given Values:
-
Apply the Formula:
-
Solve for Lattice Energy ():
Thus, the lattice energy of NaCl is -787 kJ/mol. The negative sign indicates that energy is released when the lattice is formed.
Concept Assessment Exercise 6.5
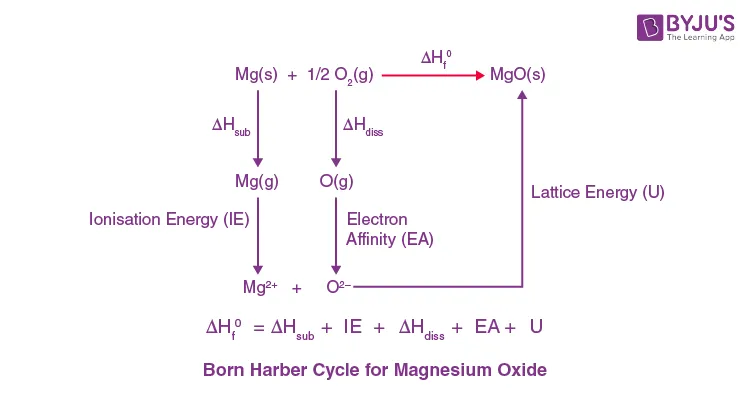
Q: Draw a complete Born-Haber Cycle for the formation of and calculate its lattice energy from the following data.
| Enthalpy Change | Value |
|---|---|
| Standard enthalpy of formation of MgO | |
| Standard enthalpy of sublimation of Mg | |
| Total Ionization energy of Mg to | |
| Standard enthalpy of atomization of () | |
| Total Electron affinity of O to (EA₁ + EA₂) |
A: The steps are:
Using Hess's Law:
Factors Affecting Lattice Energy
Lattice energy is influenced by the charge density of the ions involved. Two main factors determine the magnitude of lattice energy:
-
Ionic Charge: As the magnitude of the ionic charges increases, the electrostatic attraction between ions becomes stronger, leading to a higher (more exothermic) lattice energy.
- Example: The lattice energy of ( and ) is significantly higher than that of ( and ) because of the greater charges.
-
Ionic Radius: As the ionic radius increases, the distance between the centers of the ions increases. This weakens the electrostatic attraction, leading to a lower (less exothermic) lattice energy.
- Example: Both and have ions with +1 and -1 charges. However, the ion is larger than the ion. Therefore, has a higher lattice energy than .
Concept Assessment Exercise 6.6
Q: Which of the following compounds is expected to have higher lattice energy? Why? a. LiF or NaF b. NaCl or
A: a. LiF has a higher lattice energy. Both have a -1 fluoride ion, but the ion is smaller than the ion, resulting in a stronger attraction. b. has a higher lattice energy. The magnesium ion has a +2 charge (), which creates a much stronger electrostatic attraction with the chloride ions compared to the +1 charge of the sodium ion () in NaCl.
Role of Hydration in the Dissolving Process
When an ionic solute dissolves in a solvent like water, two main energy changes occur:
- Breaking the Lattice: Energy is required to overcome the ionic attractions and separate the ions from the crystal lattice. This is an endothermic process, equal in magnitude but opposite in sign to the lattice energy ().
- Hydration of Ions: Energy is released when the separated gaseous ions are surrounded and stabilized by solvent molecules (water). This is an exothermic process known as the enthalpy of hydration ().
The overall energy change, the enthalpy of solution (), is the sum of these two steps.
- If is negative (exothermic), the solution gets warmer. This occurs when the hydration energy released is greater than the energy required to break the lattice.
- Example:
- If is positive (endothermic), the solution gets colder. This occurs when the energy required to break the lattice is greater than the hydration energy released.
- Example:
| Substance | Enthalpy of Solution () |
|---|---|
| LiCl | -37.0 |
| NaCl | +2.98 |
| KCl | +17.2 |
| KI | +20.3 |
| +25.7 | |
| -321.0 |
Concept Assessment Exercise 6.7
Q: Calculate the enthalpy of solution for NaCl from the following data:
A:
- Energy to break the lattice:
- Energy released by hydration:
- Calculate enthalpy of solution: This small positive value indicates that dissolving NaCl in water is slightly endothermic, which is consistent with experimental data.
Factors Affecting Enthalpy of Hydration
Similar to lattice energy, the enthalpy of hydration is influenced by the charge density of the ion.
-
Ionic Charge: Ions with a higher charge create a stronger ion-dipole attraction with water molecules, resulting in a more exothermic (more negative) enthalpy of hydration.
- Example: The hydration enthalpy of ions is much more exothermic than that of ions.
-
Ionic Radius: Smaller ions have a higher charge density, allowing water molecules to approach more closely and form stronger ion-dipole interactions. This leads to a more exothermic enthalpy of hydration.
- Example: The ion is smaller than the ion. Therefore, the enthalpy of hydration for is more exothermic than for .
Concept Assessment Exercise 6.8
Q: Which has a higher (more exothermic) enthalpy of hydration? Justify your answer. a. LiF or NaF b. or
A: a. LiF. The cations ( vs ) are the difference. Since is smaller than , it has a higher charge density and thus a more exothermic enthalpy of hydration. b. . The anions are the same (). The ion is smaller than the ion, giving it a higher charge density and a more exothermic enthalpy of hydration.