6.4 Hess's Law
Hess's Law is a fundamental principle in thermochemistry based on the fact that enthalpy is a state function. This means the total enthalpy change for a chemical reaction is independent of the pathway taken from the initial state (reactants) to the final state (products). The change in enthalpy is the same whether the reaction occurs in a single step or in multiple sequential steps.
The law, stated by G.H. Hess in 1840, can be summarized as:
The total enthalpy change in a chemical reaction is the same regardless of whether the reaction is completed in one step or in several steps.
Mathematically, for any cyclic process, the sum of all enthalpy changes is zero:
Consider a reaction where reactant A transforms into product B.
- Direct Path: with an enthalpy change of .
- Indirect Path: with enthalpy changes , , and for each step.
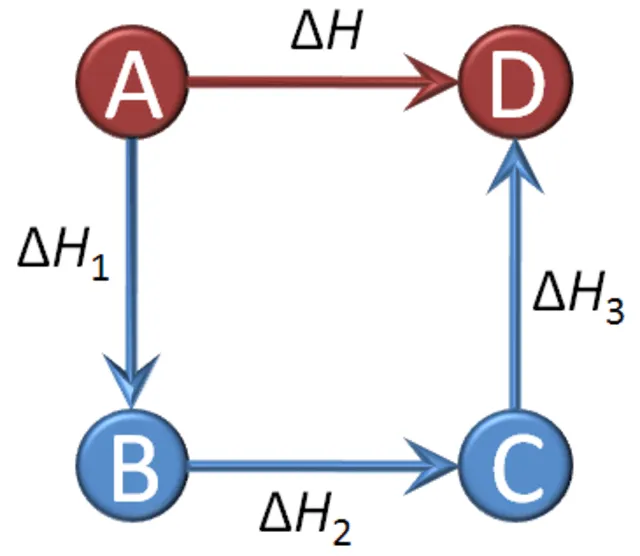
According to Hess's Law, the enthalpy change for the direct path is equal to the sum of the enthalpy changes for the indirect path:
Principle and Application
Hess's Law is particularly useful for determining the enthalpy changes of reactions that are difficult or impossible to measure directly in a calorimeter. This includes:
- Reactions that are very slow.
- Reactions that do not go to completion.
- Reactions that produce side products.
- Formation of compounds that cannot be synthesized directly from their elements (e.g., ).
- Combustion of elements that form a protective oxide layer (e.g., , ).
By using Hess's Law, we can calculate the desired enthalpy change by combining the known enthalpy changes of other related reactions.
Calculating Enthalpies of Reaction from Enthalpies of Formation ()
A common application of Hess's Law is to calculate the standard enthalpy change of a reaction () using the standard enthalpies of formation () of the reactants and products.
The general formula is:
Where:
- and are the stoichiometric coefficients of the products and reactants, respectively.
- Important Note: The standard enthalpy of formation () of any element in its most stable standard state is defined as zero.
Calculation of Enthalpy of Combustion ()
The enthalpy of combustion, , is the heat evolved when one mole of a substance burns completely in oxygen under standard conditions. It can be calculated using the same formula as above, with the products typically being and .
Worked Examples
Example 6.6: Combustion of Carbon
The complete combustion of carbon to carbon dioxide can occur in one step or two steps.
Single Step:
Two Steps:
- Formation of carbon monoxide:
- Combustion of carbon monoxide:
Applying Hess's Law:
The total enthalpy change for the two-step process is the sum of the individual steps:
This matches the enthalpy change for the single-step reaction, demonstrating the path independence.
Figure 6.4: Energy cycle for the reaction between and to produce .
Example 6.7: Enthalpy of Formation of Methane
Calculate the standard enthalpy of formation for methane () using the following combustion data.
Target Reaction:
Given Reactions:
(i)
(ii)
(iii)
Solution:
-
Manipulate the given equations to match the target reaction:
- Equation (iii): Use as is — provides as a reactant.
- Equation (ii): Multiply by 2 — provides as reactants.
- Equation (i): Reverse the reaction — places as a product. Sign of is changed:
-
Combine the manipulated equations:
\begin{align*} \mathrm{C_{(s)} + O_{2(g)}} &\rightarrow \mathrm{CO_{2(g)}} & \Delta H^{\circ} &= -393.5 \,\text{kJ} \\ \mathrm{2H_{2(g)} + O_{2(g)}} &\rightarrow \mathrm{2H_2O_{(l)}} & \Delta H^{\circ} &= -571.6 \,\text{kJ} \\ \mathrm{CO_{2(g)} + 2H_2O_{(l)}} &\rightarrow \mathrm{CH_{4(g)} + 2O_{2(g)}} & \Delta H^{\circ} &= +890.4 \,\text{kJ} \end{align*}
- Cancel species appearing on both sides (, , cancel):
- Sum the enthalpy changes:
The standard enthalpy of formation of methane is .