6.3 HEAT AND TEMPERATURE
This section explores the fundamental concepts of heat and temperature, their relationship, and how heat changes in chemical reactions are measured using calorimetry.
Heat as a Form of Energy
Heat is a form of energy that arises from the movement (kinetic energy) of molecules within a substance. When a body absorbs heat, it becomes hotter; when it releases heat, it becomes colder.
The Law of Conservation of Energy states that energy cannot be created or destroyed, only converted from one form to another. Heat is one of these forms, alongside light, mechanical, chemical, and nuclear energy.
Example: Boiling water in a pot causes the lid to jump. This demonstrates the conversion of heat energy from the stove into mechanical energy that moves the lid.
The SI unit for heat is the Joule (J). It is also commonly measured in calories (cal).
Heat vs. Temperature
While related, heat and temperature are distinct concepts. Temperature is considered an intensive property because it does not depend on the amount of matter present.
| Feature | Heat () | Temperature () |
|---|---|---|
| Definition | The total energy produced by the movement of all molecules in a substance. | A measure of the average kinetic energy of the molecules in a substance. It describes the intensity of heat (hotness or coldness). |
| Type of Property | Extensive (depends on amount of matter) | Intensive (independent of amount of matter) |
| Depends On | Particle speed, particle size, and number of particles. | Particle speed (average kinetic energy) only. |
| Units | Joules (J), calories (cal) | Kelvin (K), Celsius (°C), Fahrenheit (°F) |
Example: A large jar and a small glass both filled with water at the same temperature. The water in both has the same average kinetic energy (same temperature), but the jar contains more water molecules, so it possesses more total heat energy.
Heat Capacity and Specific Heat Capacity
The amount of heat absorbed () by a substance is directly proportional to its temperature change ().
Heat Capacity ()
The amount of heat required to raise the temperature of a given amount of a substance by 1 Kelvin.
- Units: Joules per Kelvin ()
- Heat capacity is an extensive property (depends on the amount of substance).
Specific Heat Capacity ()
The amount of heat required to raise the temperature of one gram of a substance by 1 Kelvin.
This leads to the fundamental equation for heat calculation:
Where:
- = heat absorbed or released (J)
- = mass of the substance (g)
- = specific heat capacity ()
- = change in temperature (K or °C)
Molar Heat Capacity
The heat capacity per mole of a substance ().
Where is the molar mass in .
Example:
- Specific heat capacity of Copper (Cu) =
- Molar mass of Cu =
- Molar heat capacity of Cu =
Measurement of Heat of Reaction (Calorimetry)
A calorimeter is a device used to measure the heat flow associated with a chemical or physical process.
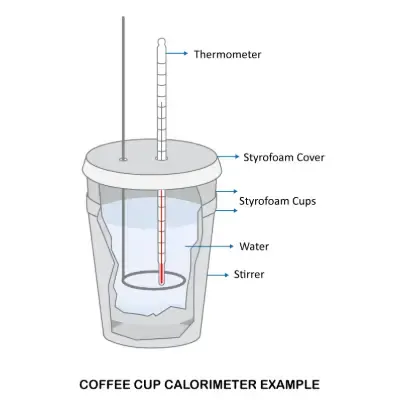
Constant Pressure Calorimeter (Coffee Cup Calorimeter)
This type of calorimeter measures heat changes at constant atmospheric pressure. A simple and common version is the coffee cup calorimeter. It consists of:
- A thermally insulated container (e.g., Styrofoam cup)
- A lid
- A thermometer
- A stirrer
The heat absorbed or released by the reaction () is equal in magnitude but opposite in sign to the heat absorbed or released by the solution ():
The heat change of the solution is calculated using:
Where is the total mass of the solution and is the specific heat of the solution (often assumed to be that of water = ).
Worked Examples
Example 6.4: Calculating Heat of Neutralization
When of NaOH neutralizes of HCl at , the temperature rises to . Calculate the heat of neutralization. (Specific heat of water = . Density of water = )
Solution:
Step 1: Identify given values
- Volume of NaOH =
- Volume of HCl =
- Total Volume =
- Density of solution =
- Specific heat capacity () =
- Initial Temperature () =
- Final Temperature () =
Step 2: Calculate the mass () of the reaction mixture
Step 3: Calculate the change in temperature ()
Step 4: Calculate the heat evolved () by the solution
Step 5: Calculate the heat of reaction ()
The negative sign indicates the reaction is exothermic (heat is released by the reaction into the solution).
Step 6: Calculate moles of water formed
The neutralization reaction is:
Moles of NaOH =
Moles of HCl =
Moles of formed =
Step 7: Calculate heat of neutralization per mole
This value is close to the standard heat of neutralization of a strong acid with a strong base ().