2.24 Chemical Nature and Role of ATP and NAD
This section details the structure and biological function of two critical molecules in cellular metabolism: Adenosine Triphosphate (ATP) and Nicotinamide Adenine Dinucleotide (NAD).
Adenosine Triphosphate (ATP)
ATP is a mononucleotide that serves as the primary energy carrier in all living cells. It is synthesized within organelles like mitochondria and chloroplasts.
Ultrastructure of Animal and Plant Cells→
Structure
ATP is composed of three main parts linked by covalent bonds:
- Adenine: A nitrogenous base.
- Ribose: A five-carbon sugar.
- Three Phosphate Groups: Linked in a chain. The two covalent bonds connecting the terminal phosphate groups are called high-energy bonds (represented by the symbol ~). These are technically phosphoanhydride bonds.
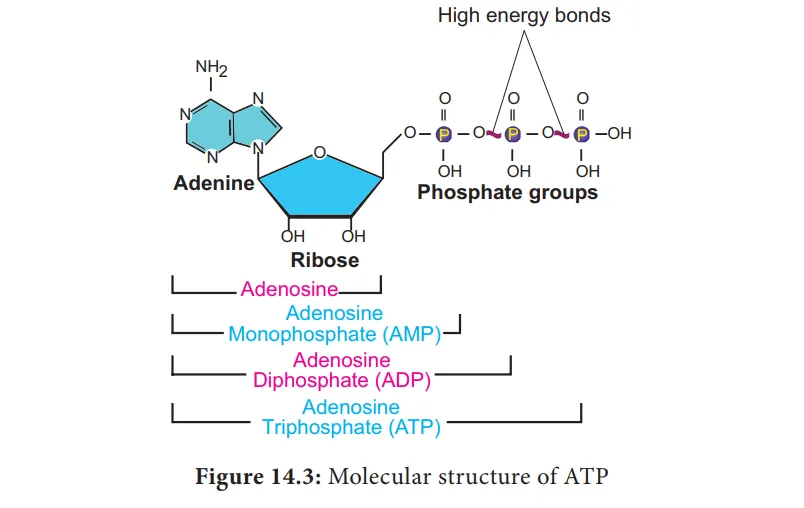
Function
- ATP is known as the energy currency of the cell.
- Energy is released when ATP is hydrolyzed (broken down with water) into Adenosine Diphosphate (ADP) and an inorganic phosphate ():
- The hydrolysis of one mole of ATP releases approximately 7.3 kcal/mol (30.6 kJ/mol) of energy under standard conditions.
- ATP is regenerated from ADP and by the process of phosphorylation, which requires energy input from cellular respiration or photosynthesis.
Nicotinamide Adenine Dinucleotide (NAD)
NAD is a dinucleotide that functions as a coenzyme, primarily involved in redox reactions (electron transfer). It is classified as a coenzyme — a type of organic cofactor that assists enzymes.
Structure
NAD consists of two nucleotides joined by their phosphate groups:
- Nucleotide 1: Composed of nicotinamide (a derivative of Vitamin B3/Niacin), a ribose sugar, and a phosphate group.
- Nucleotide 2: Composed of adenine, a ribose sugar, and a phosphate group.
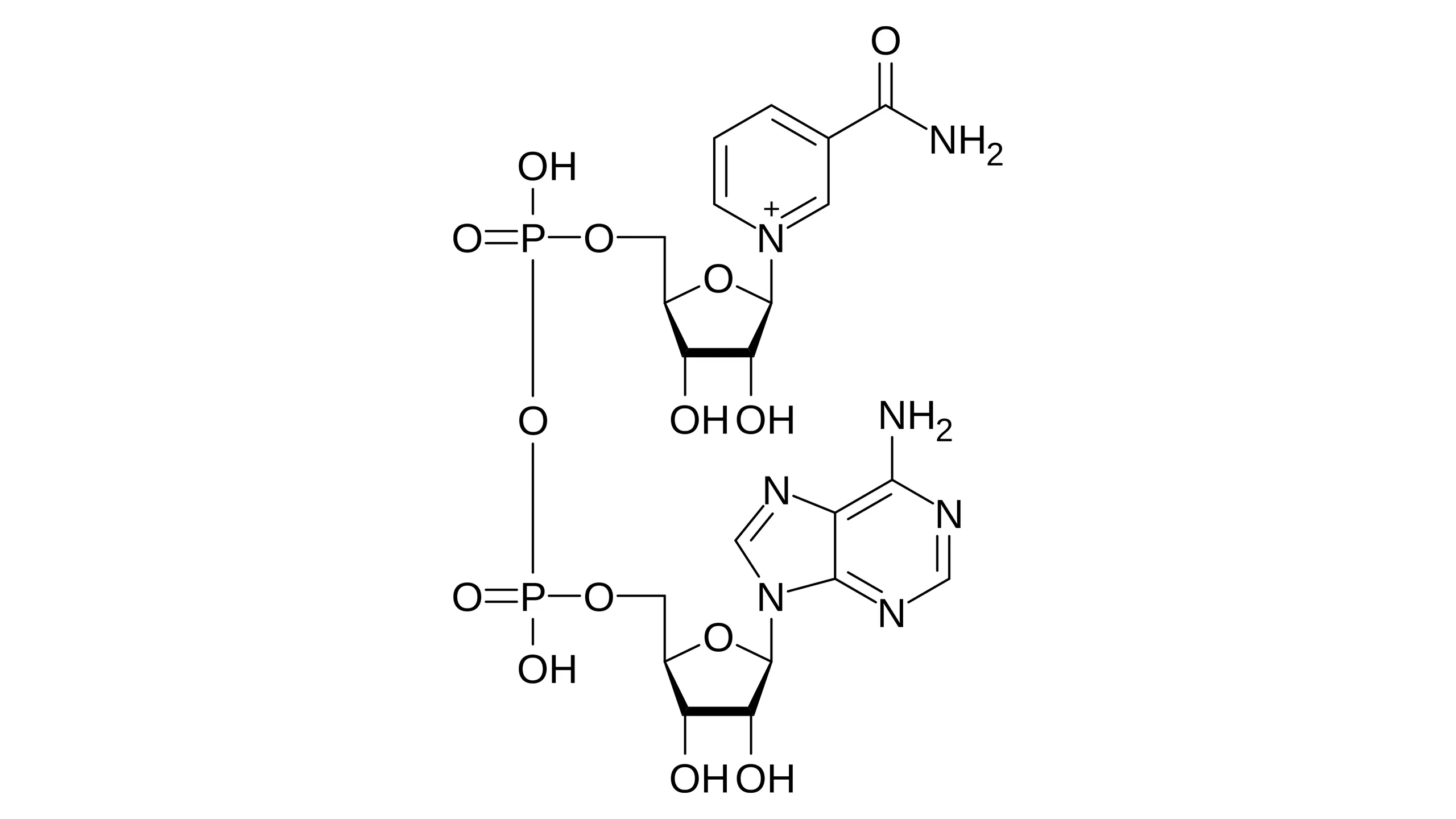
Function
- NAD acts as a coenzyme, an essential organic molecule that helps enzymes function correctly.
- It plays a crucial role in cellular respiration by accepting and donating electrons (hydrogen atoms).
- Oxidized form: — acts as an electron acceptor.
- Reduced form: — formed after accepting two electrons and one proton; donates electrons to the Electron Transport Chain.
Biological Significance
Both ATP and NAD are fundamental to life, linking energy-releasing (catabolic) and energy-requiring (anabolic) processes:
| Feature | ATP | NAD |
|---|---|---|
| Type | Mononucleotide | Dinucleotide |
| Role | Direct energy currency | Electron/hydrogen carrier |
| Key reaction | Hydrolysis → ADP + | ↔ |
| Vitamin precursor | — | Niacin (Vitamin B3) |
ATP provides the direct energy for cellular work (active transport, muscle contraction, biosynthesis), while NAD facilitates the transfer of energy from food molecules to the process of ATP synthesis.