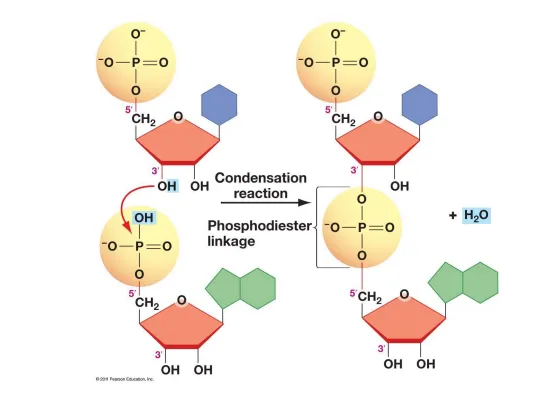
2.23 Formation of Phosphodiester Bond and Polymerization of Nucleotides
The process by which individual nucleotides are linked together to form a long chain, known as a polynucleotide (like DNA or RNA), is a fundamental process in the synthesis of genetic material.
Key Concepts
- Reaction Type: The joining of nucleotides is a condensation reaction.
- Bond Formed: A phosphodiester bond is created between two adjacent nucleotides.
- Molecule Released: Unlike many other biological condensation reactions that release water, this process releases pyrophosphate (), which consists of two phosphate groups linked together.
- Directionality: The resulting polynucleotide chain has a distinct directionality, with a free 5' phosphate group at one end and a free 3' hydroxyl (OH) group at the other. By convention, nucleotide sequences are read from the 5' to the 3' end.
Mechanism of Polymerization
The formation of the phosphodiester bond occurs in a series of steps, driven by the energy from an incoming nucleotide.
-
Initiation: The process requires a pre-existing polynucleotide chain with a free 3' hydroxyl group and an incoming nucleotide triphosphate (NTP). The NTP provides both the next nucleotide and the energy for the reaction.
-
Bond Formation:
- The free 3' hydroxyl group of the last nucleotide in the growing chain attacks the innermost (alpha) phosphate group of the incoming NTP.
- A phosphodiester bond is formed, linking the 5' carbon of the new nucleotide to the 3' carbon of the previous nucleotide.
- As the bond forms, the bond between the alpha and beta phosphates of the NTP is broken.
-
Pyrophosphate Release & Hydrolysis:
- A molecule of pyrophosphate () is released.
- This pyrophosphate is then quickly hydrolyzed (split by adding water) into two individual phosphate ions ().
- The hydrolysis of pyrophosphate releases a significant amount of energy, which makes the overall polymerization reaction irreversible and drives it forward to completion.
Summary
- Nucleotides are joined together by phosphodiester bonds to form polynucleotide chains (DNA and RNA).
- This is a condensation reaction that releases pyrophosphate (), not water.
- The incoming monomer is a nucleotide triphosphate (NTP), which provides both the nucleotide and the energy.
- The hydrolysis of pyrophosphate is an energy-releasing step that drives the polymerization reaction forward.
- The resulting chain is directional, with a 5' end (free phosphate) and a 3' end (free hydroxyl group).
This process is biologically significant as it is the basis for DNA replication and transcription (RNA synthesis), essential for storing and expressing genetic information in all living organisms. See also Nucleic Acid→ and Central Dogma of Life→.