8.8 Kinetic Theory of Gases
The Kinetic Theory of Gases is a scientific model that explains the macroscopic properties of a gas, such as pressure, volume, and temperature, by considering the motion of its constituent molecules. Developed in the 19th century by scientists like Maxwell and Clausius, the theory describes a gas as a large number of submicroscopic particles (atoms or molecules) that are in constant, rapid, random motion.
The Microscopic Model of a Gas
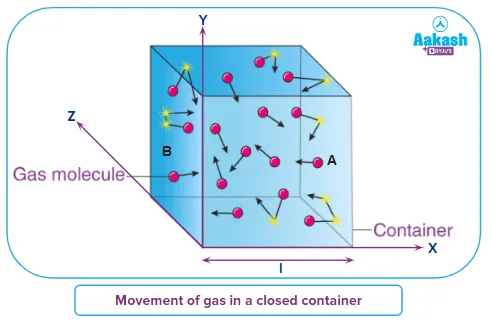
The core idea of the kinetic theory is that a gas is composed of a vast number of tiny, hard spheres (molecules) that are in continuous, chaotic motion. These molecules collide with each other and with the walls of their container.
Assumptions of the Kinetic Theory for an Ideal Gas
To simplify the model, the theory makes several key assumptions:
- Large Number of Molecules: The gas consists of a very large number of identical molecules.
- Negligible Molecular Volume: The size of the molecules is negligible compared to the average distance between them.
- Random Motion: The molecules are in constant, random motion, following Newton's laws.
- Elastic Collisions: All collisions between molecules and with the container walls are perfectly elastic, meaning kinetic energy is conserved.
- No Intermolecular Forces: Molecules do not exert any long-range forces on each other; they only interact during collisions.
- Negligible Collision Time: The time spent during a collision is negligible compared to the time between collisions.
Linking Microscopic and Macroscopic Properties
The power of the kinetic theory is its ability to connect the macroscopic properties we can measure with the microscopic behavior of molecules.
-
Pressure (): The pressure of a gas is the result of the countless collisions of its molecules with the walls of the container. From kinetic theory: where is the gas density and is the mean square speed.
-
Temperature (): The absolute temperature of a gas is directly proportional to the average translational kinetic energy of its molecules: where J/K is the Boltzmann constant. Higher temperature means higher average molecular speed.
-
Volume (): At constant temperature and pressure, increasing the number of molecules increases the volume. At constant volume, increasing temperature increases pressure (more frequent, harder collisions).
Root Mean Square (RMS) Speed
The RMS speed is a useful measure of the typical molecular speed: where J mol K is the universal gas constant, is absolute temperature, and is the molar mass. Heavier gases have lower RMS speeds at the same temperature.
The Ideal Gas Law
The relationship between pressure, volume, temperature, and the amount of gas is described by the Ideal Gas Law:
Where:
- = Pressure of the gas (Pa)
- = Volume of the gas (m³)
- = Number of moles of the gas
- = Universal gas constant ()
- = Absolute temperature (K)
An equivalent form using the number of molecules and Boltzmann constant :
An ideal gas is a theoretical gas that perfectly follows these assumptions. A real gas behaves most like an ideal gas at low pressure and high temperature, where molecules are far apart and intermolecular forces are negligible.
Why do real gases deviate? At high pressure, molecular volume is no longer negligible. At low temperature, intermolecular attractive forces become significant.
Density of an Ideal Gas
From and : Density is directly proportional to pressure and molar mass, and inversely proportional to temperature.
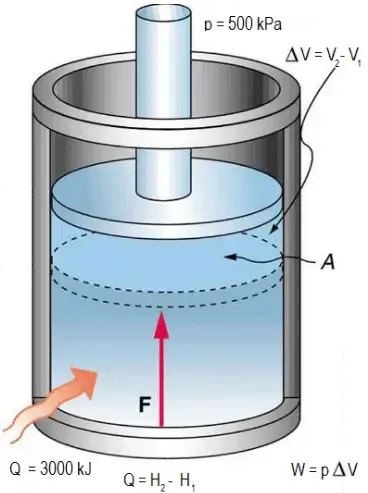
Work in Thermodynamics
In thermodynamics, work is a form of energy transfer. For a gas in a cylinder with a movable piston, work is done when the gas expands or is compressed.
Work Done at Constant Pressure
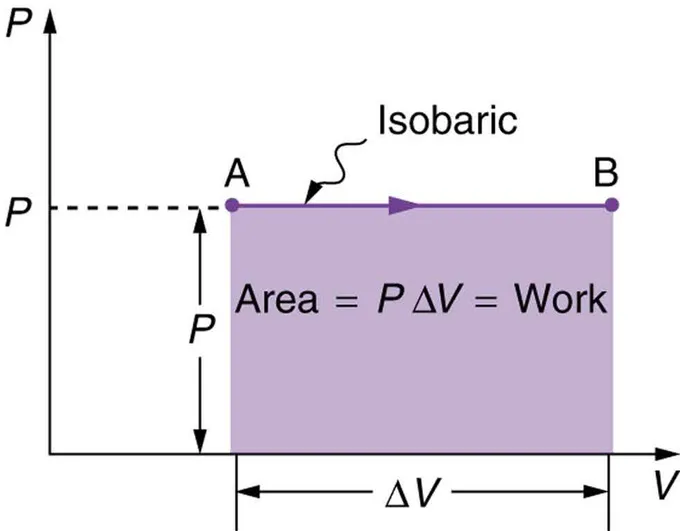
When a gas expands or contracts at constant pressure , the work done is:
where is the change in volume.
Work Done by the Gas vs. Work Done on the Gas
| Situation | Work | Sign | |
|---|---|---|---|
| Gas expands (pushes piston out) | Done by the gas on surroundings | Positive () | |
| Gas compressed (piston pushed in) | Done on the gas by surroundings | Negative () |
Sign Convention:
- Work done by the gas (expansion): positive ()
- Work done on the gas (compression): negative ()
The Pressure-Volume (P-V) Graph
The work done during a thermodynamic process can be represented graphically as the area under the curve on a P-V diagram.