8.7 Heat Engine
A heat engine is a device designed to convert thermal energy (heat) into mechanical work. It operates by absorbing heat from a high-temperature source, converting a portion of that heat into useful work, and rejecting the remaining energy as waste heat to a lower-temperature sink. This process is fundamental to many technologies, from power plants to vehicle engines.
1. Components of a Heat Engine
Every heat engine, regardless of its specific design, consists of three essential components:
| Component | Role | Description |
|---|---|---|
| Hot Reservoir | Heat Source | A body at a high temperature () that supplies heat energy () to the engine. |
| Working Substance | Conversion Medium | A fluid (like a gas or steam) that absorbs heat, expands, and does mechanical work. |
| Cold Reservoir | Heat Sink | A body at a lower temperature () that absorbs the waste heat () rejected by the engine. |
Figure 1: Heat Engine Components
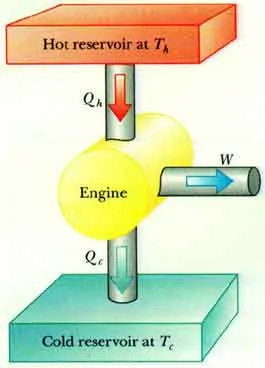
2. Working Principle and Efficiency
A heat engine operates in a continuous cycle. In each cycle:
- Heat () is absorbed from the hot reservoir.
- The working substance expands, performing mechanical work ().
- Waste heat () is expelled to the cold reservoir.
The thermal efficiency () of a heat engine is the ratio of the useful work done to the heat energy supplied.
According to the First Law of Thermodynamics, the work done is the difference between the heat absorbed and the heat rejected (). Therefore, the efficiency is:
The Kelvin-Planck statement of the Second Law of Thermodynamics dictates that no heat engine can be 100% efficient; some waste heat () must always be rejected.
Application: The Petrol Engine
The petrol engine (or gasoline engine) is a common type of internal combustion engine used in most cars. It is a prime example of a heat engine.
- Function: It burns a mixture of petrol and air inside its cylinders to create high-temperature, high-pressure gas. This gas expands and pushes pistons, generating mechanical work.
- Efficiency: Petrol engines are not very efficient. Typically, only 25% to 30% of the chemical energy in the fuel is converted into useful work. The rest is lost as heat through the exhaust and cooling system.
The Four-Stroke Cycle
Most petrol engines operate on a four-stroke cycle:
| Stroke | Piston Movement | Valves | Action |
|---|---|---|---|
| 1. Intake | Down | Inlet open, Outlet closed | The piston moves down, drawing a fuel-air mixture into the cylinder. |
| 2. Compression | Up | Both closed | The piston moves up, compressing the fuel-air mixture adiabatically. |
| 3. Power | Down | Both closed | A spark plug ignites the mixture, causing a rapid expansion that pushes the piston down, doing work. |
| 4. Exhaust | Up | Inlet closed, Outlet open | The piston moves up, pushing the burnt gases out of the cylinder. |
Figure 2: Petrol Engine Strokes
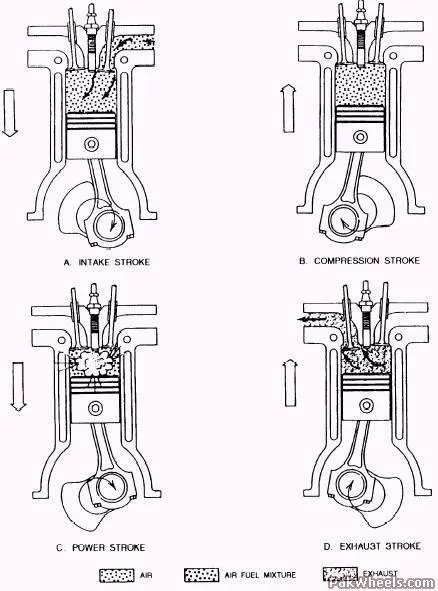
Carnot Engine
A Carnot engine is a theoretical, ideal heat engine that operates on the Carnot cycle — a reversible cycle consisting of four processes:
- Isothermal expansion at temperature (heat absorbed from hot reservoir)
- Adiabatic expansion (temperature drops from to )
- Isothermal compression at temperature (heat rejected to cold reservoir)
- Adiabatic compression (temperature rises from back to )
Carnot Efficiency
The efficiency of a Carnot engine depends only on the absolute temperatures of the hot source () and cold sink ():
where temperatures are in Kelvin.
Carnot's Theorem (Efficiency Limit)
Carnot's Theorem states that:
- No heat engine operating between two given temperatures can be more efficient than a Carnot engine operating between the same temperatures.
- All reversible engines operating between the same two temperatures have the same efficiency.
- The Carnot efficiency sets the upper limit for the efficiency of any real heat engine.
This is a direct consequence of the Second Law of Thermodynamics. Since always, the Carnot efficiency is always less than 1 (i.e., less than 100%).
Example: A Carnot engine operating between and has efficiency:
Possible Questions and Answers
-
Q: Why can't a heat engine be 100% efficient?
A: This is a consequence of the Second Law of Thermodynamics. To operate in a continuous cycle, an engine must return to its initial state. To do this, it must expel waste heat to a cold reservoir. It is fundamentally impossible to convert all the absorbed heat into work without rejecting some of it.
-
Q: What is the difference between an internal and an external combustion engine?
A: In an internal combustion engine (like a petrol engine), the fuel is burned inside the engine's cylinders, and the hot gases produced are the working substance. In an external combustion engine (like a steam engine), the fuel is burned outside the engine to heat a separate working substance (like water/steam), which then does the work.
-
Q: How does the Carnot efficiency formula show that 100% efficiency is impossible?
A: The formula shows that for , we would need (absolute zero), which is unattainable. Therefore, always.