20.5 X-Ray Crystallographic Analysis
20.5.1 Introduction to X-ray Crystallography
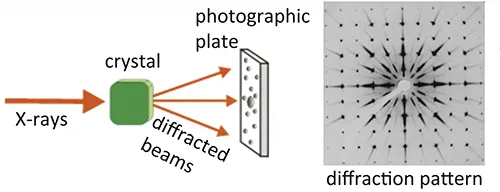
X-ray crystallography is a powerful analytical technique that utilizes X-rays to determine the detailed atomic and molecular structure of a crystal. This method reveals:
- The precise arrangement of atoms and molecules within the crystal lattice.
- Information about how these atoms and molecules are bonded together.
- Any irregularities or defects present in the crystal structure.
This technique is versatile and can be applied to a wide range of substances, including:
- Inorganic compounds: Salts, minerals, metals, semiconductors.
- Biological macromolecules: Proteins, DNA, RNA, vitamins.
X-ray crystallography is an invaluable tool across various scientific disciplines, such as:
- Biology: Understanding the structure and function of biomolecules.
- Chemistry: Elucidating the structure and reactivity of compounds.
- Geology: Studying mineral structures and geological processes.
Interesting Fact: Barium Tests Barium tests use Barium Sulphate (), a white, chalky substance, to examine the digestive tract. Patients drink this substance before an X-ray, which allows doctors to visualize the internal structures and make accurate diagnoses.
20.5.2 Applications of X-ray Crystallography
X-ray crystallography has played a crucial role in numerous groundbreaking scientific discoveries and continues to be fundamental in various fields. Some notable historical achievements include determining the structures of simple crystals like quartz and salt. A landmark achievement was the elucidation of the double-helix structure of DNA by Rosalind Franklin, James Watson, and Francis Crick in 1953. Other significant molecules analyzed include Vitamin B12, insulin, and penicillin.
Key applications of X-ray crystallography include:
-
Understanding Biomolecular Structures:
- Determining the 3D structures of proteins and other biomolecules, which is essential for understanding their biological functions.
- Revealing the structures of DNA and RNA, providing insights into genetic information storage and transmission mechanisms.
-
Drug Discovery and Design:
- By identifying the structure of target proteins (e.g., enzymes, receptors), it helps in locating potential binding sites for new drugs, facilitating rational drug design.
-
Materials Science and Engineering:
- Understanding the atomic structure of various materials (metals, ceramics, polymers) to develop new materials with desired properties (e.g., strength, conductivity).
- Studying the structure of nanoparticles to advance nanotechnology applications.
-
Fundamental Chemistry:
- Determining the structure of complex organic and inorganic compounds, which is crucial for understanding their reactivity, stability, and physical properties.
- Investigating intermediates and transition states in chemical reactions to elucidate reaction mechanisms. For more on analytical techniques, see IR Spectroscopy→.
-
Geology and Mineralogy:
- Determining the structures of minerals to understand geological processes and the formation of natural resources.
- Studying how minerals transform under different temperature and pressure conditions.
-
Physical Properties of Solids:
- Explaining various physical properties (e.g., electrical conductivity, magnetism, optical properties) by understanding the arrangement of atoms in solid-state materials.
-
Environmental Science:
- Understanding the crystalline structures of pollutants to aid in developing effective methods for their removal or neutralization.