18.3 IR Spectroscopy
Infrared (IR) spectroscopy is an analytical technique that utilizes the interaction of infrared radiation with organic molecules. It is primarily used to identify the functional groups present in a compound. The typical range of wavenumbers for IR spectroscopy is from to .
This technique is widely applied to both organic and inorganic compounds for the identification of molecular structures. Its high scan speed, resolution, and sensitivity make it an invaluable analytical tool.
IR spectroscopy is complementary to other spectroscopic techniques and helps in determining the index of hydrogen deficiency or unsaturation of organic molecules.
18.3.1 Principle of IR Spectroscopy
The fundamental principle of IR spectroscopy involves the interaction between the electric field of infrared radiation and the dipole moment of a molecule.
- When a molecule is exposed to IR radiation, it can absorb energy if the frequency of the radiation matches the natural vibrational frequency of one of its chemical bonds.
- This absorption of energy causes the bond to vibrate with a greater amplitude.
- For a bond to absorb IR radiation, its vibration must cause a change in the bond's dipole moment.
- The intensity of the absorption is proportional to the polarity of the bond; more polar bonds produce stronger absorptions.
The resulting IR spectrum shows the frequencies of absorbed radiation, which are characteristic of the molecular structure and the specific functional groups present in the sample.
Major Components of an IR Spectrophotometer:
- Source of IR radiations
- Monochromator (to select specific frequencies)
- Sample chamber
- Detector
- Data collection and processing system
18.3.2 Reading an IR Spectrum
Interpreting an IR spectrum involves analyzing the various absorption bands (peaks) to identify functional groups.
- Each peak in the spectrum corresponds to the vibration of a specific bond. This creates a unique fingerprint for each compound.
- The analysis begins by correlating the absorption frequencies of the unknown compound with reference charts that list characteristic wavenumbers for different types of bonds.
- Key functional groups like O-H, N-H, C-H, and C=O have very distinct and strong absorption peaks, making them easy to identify.
- The fingerprint region, located below , contains a complex pattern of peaks that is unique to each molecule, aiding in the definitive identification of a compound by comparison to a known spectrum.
18.3.3 Common Functional Groups and Their Characteristic IR Absorption Ranges
The following table summarizes the characteristic absorption ranges for common functional groups.
Table 18.1: Functional Groups with Wavenumber and Intensity
| Functional Group | Bond Type | Wavenumber () | Intensity |
|---|---|---|---|
| Alkyl | C-H | 2853-2962 | medium-strong |
| Alkenyl | =C-H | 3010-3095 | medium |
| C=C | 1620-1680 | variable | |
| Alkynyl | ≡C-H | ~3300 | strong |
| C≡C | 2100-2260 | variable | |
| Aromatic | Ar-H | ~3030 | variable |
| C=C (ring) | 1400-1600 | variable | |
| Alcohols, Phenols | O-H | 3200-3500 | strong, broad |
| C-O | 1025-1060 | strong | |
| Carboxylic Acids | O-H | 2500-3000 | very broad, variable |
| C=O | 1710-1780 | strong | |
| Aldehydes | C=O | 1690-1740 | strong |
| Ketones | C=O | 1680-1750 | strong |
| Esters | C=O | 1735-1750 | strong |
| Amides | C=O | 1630-1690 | strong |
| Amines | N-H | 3300-3500 | medium |
Worked Examples
Example 18.2: Interpret the IR spectrum of ethanol ()
Problem Solving Strategy:
- Identify the significant peaks in the provided spectrum.
- Compare the wavenumbers of these peaks to the known ranges for various functional groups (from the reference table).
- Deduce the presence of specific functional groups based on the matches.
- Confirm the structure of the molecule.
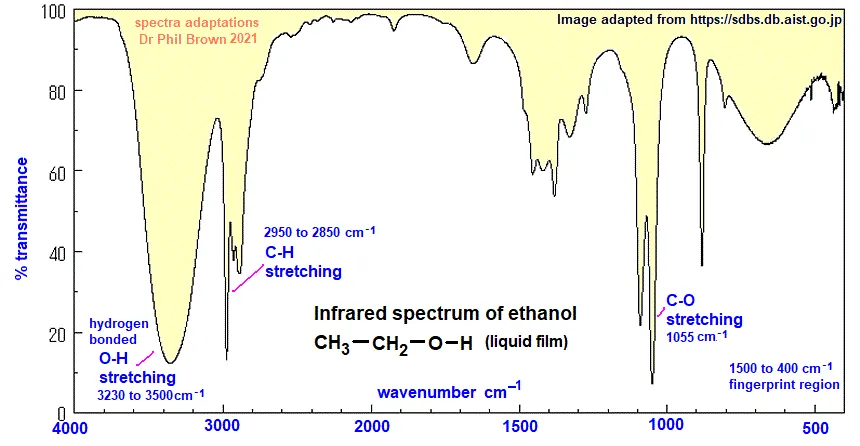
Observed Peaks in Ethanol Spectrum:
- A very broad peak around
- Peaks in the range of
- A strong peak around
Solution (Matching Peaks to Functional Groups):
- Broad Peak at : This characteristic broad absorption strongly suggests the presence of an O-H bond from an alcohol functional group. The broadness is due to hydrogen bonding.
- Peaks at : These peaks are characteristic of C-H single bond stretching vibrations from an alkyl group (the ethyl group, ).
- Peak at : This absorption corresponds to the C-O single bond stretching vibration, further confirming the presence of an alcohol group.
IR Spectrum of Acetone ()
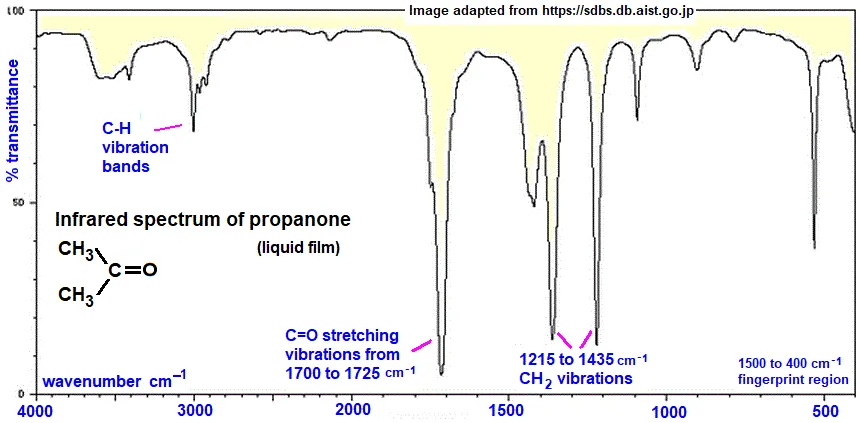
- Strong peak at : This is a very strong, sharp peak characteristic of the C=O (carbonyl) functional group in a ketone. This peak is also found in aldehydes, carboxylic acids, and their derivatives, though the exact wavenumber varies slightly.
- Peaks near : These are the C-H stretching vibrations from the methyl groups ().
- Vibrations at : These correspond to bending vibrations.
IR Spectrum of Phenol ()
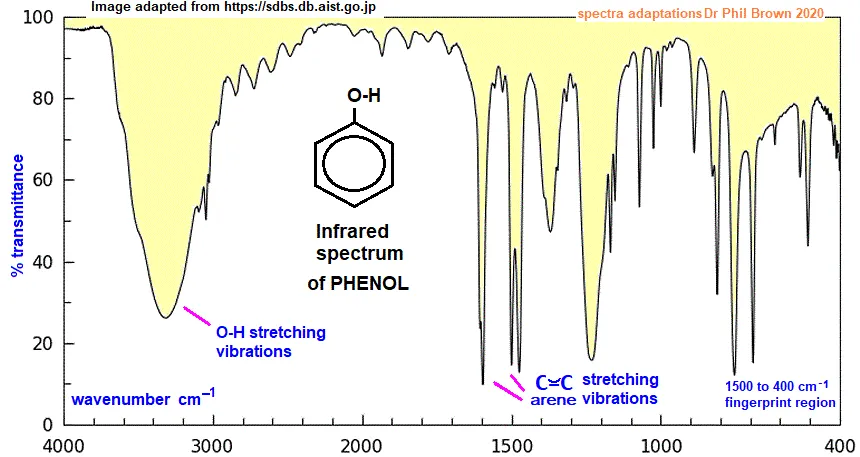
- Broad, strong peak around : This represents the O-H functional group. As in ethanol, the peak is broadened by hydrogen bonding.
- Peaks around : These absorptions are due to the C=C double bonds within the conjugated aromatic ring.
- Peaks above : These correspond to the Ar-H (aromatic C-H) bond stretching vibrations.