18.4 UV-Visible Spectroscopy
1. Introduction to UV-Visible Spectroscopy
UV-Visible (UV-Vis) Spectroscopy is an analytical technique that measures the absorption of ultraviolet (UV) and visible light by a substance.
- Wavelength Range: The typical range is from 200 nm to 800 nm.
- Principle: The technique is based on the excitation of electrons to higher energy levels when a molecule absorbs photons of UV or visible light. The amount of light absorbed is directly proportional to the concentration of the absorbing species in the sample.
- Applications: It is widely used in material sciences and biochemistry for both qualitative and quantitative analysis. It can be applied to liquids, solids, and gases and is particularly useful for identifying conjugation in unsaturated organic compounds.
2. Electronic Transitions
The absorption of specific wavelengths of light causes electrons to transition between energy levels. The main types of electronic transitions are:
- (sigma to sigma star): High-energy transition, typically occurs in the far UV region (< 200 nm).
- (n to sigma star): Transition of a non-bonding electron to an anti-bonding sigma orbital.
- (pi to pi star): Excitation of an electron from a bonding orbital to an anti-bonding orbital. Common in compounds with double/triple bonds and aromatic rings.
- (n to pi star): Transition of a non-bonding electron to an anti-bonding orbital. Occurs in molecules with lone pairs on atoms adjacent to a -system (e.g., carbonyls).
These transitions provide valuable information about a molecule's structure.
3. Color and Wavelength Absorption
Colored compounds absorb light in the visible region of the electromagnetic spectrum (400 nm - 800 nm). The color we perceive is not the color that is absorbed, but rather its complementary color, which is the light that is transmitted or reflected.
- Energy and Wavelength: The energy required for an electronic transition is related to the wavelength of light absorbed. Shorter wavelengths (e.g., violet, blue) correspond to higher energy transitions, while longer wavelengths (e.g., orange, red) correspond to lower energy transitions.
The relationship between absorbed and observed color is shown below.
| Wavelength Absorbed (nm) | Color Absorbed | Color Observed |
|---|---|---|
| 400 - 435 | Violet | Yellow-Green |
| 435 - 480 | Blue | Yellow |
| 480 - 490 | Green-Blue | Orange |
| 490 - 500 | Blue-Green | Red |
| 500 - 560 | Green | Purple |
| 560 - 580 | Yellow-Green | Violet |
| 580 - 595 | Yellow | Blue |
| 595 - 605 | Orange | Green-Blue |
| 605 - 700 | Red | Blue-Green |
4. Predicting UV-Visible Absorption
A compound will likely absorb in the UV-visible region if its electronic structure allows for or transitions. Key structural features include:
- Chromophore: A functional group that absorbs UV or visible radiation (e.g., C=C, C=O, aromatic rings).
- Auxochrome: A functional group that does not absorb radiation itself but increases the absorption of a chromophore and shifts the absorption to a longer wavelength (e.g., -OH, -NH₂).
- Conjugated double bonds (alternating single and double bonds).
Examples
-
Benzene ()
- Has a conjugated aromatic system.
- Exhibits transitions, absorbing in the UV region around 254 nm.
-
1,3-Butadiene ()
- Has a conjugated system of two double bonds ().
- Shows transitions, absorbing in the UV region around 217 nm.
-
Acetone ()
- Contains a carbonyl group (), which has both a -bond and lone pairs on the oxygen atom.
- Can undergo transitions, absorbing in the UV region around 279 nm.
5. Beer-Lambert Law
The quantitative aspect of UV-Vis spectroscopy is governed by the Beer-Lambert Law, which states that the absorbance () of a solution is directly proportional to its concentration () and the path length ().
Where is the molar absorptivity.
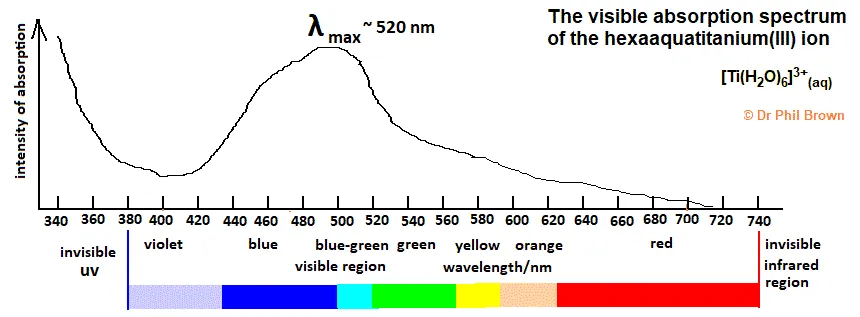
Example 1: The Color of Hexaaqua Titanium(III)
The complex ion hexaaqua Titanium(III), , is violet. This is because the complex absorbs light in the yellow-green region of the visible spectrum. The color we see (violet) is the complementary color of the light absorbed.
Example 2: Quantitative Analysis
UV-Visible Spectroscopy can be used to determine the concentration of an unknown solution.
- Create a Standard Curve: Prepare several solutions of a substance with known concentrations (standards). Measure the absorbance of each standard at a specific wavelength. Plot absorbance vs. concentration to create a calibration or standard curve.
- Measure the Unknown: Measure the absorbance of the solution with the unknown concentration at the same wavelength.
- Determine Concentration: Use the standard curve to find the concentration that corresponds to the absorbance of the unknown sample.
Possible Questions/Answers
-
Q: A compound absorbs light with a wavelength of 500 nm - 560 nm. What colour do you expect for this compound? A: According to the complementary color table, absorption in the 500-560 nm range (green light) means the compound will appear Purple.
-
Q: What wavelength do you suggest about the absorption of light by the components of air? A: The main components of air (, , Ar) are colorless. This means they do not absorb light in the visible region (400-800 nm). They absorb high-energy radiation in the far-UV region, at wavelengths below 200 nm.