20.3 Mechanism of Catalysts
This section explores the fundamental concept of catalysis, its underlying mechanisms, and the crucial role catalysts play in altering the rate of chemical reactions without being consumed themselves.
20.3.1 What is Catalysis?
Catalysis is a process where the speed of a chemical reaction is altered by a substance known as a catalyst. Catalysts are unique because they are:
- Not consumed during the reaction: They participate in the reaction mechanism but are regenerated in their original form at the end, allowing them to be used repeatedly.
- Effective in small amounts: Even a tiny quantity of a catalyst can significantly impact the reaction rate.
- Form temporary intermediates: Catalysts often form an unstable substance temporarily during the reaction before returning to their original state, enabling a cyclical catalytic process.
Effect on Reaction Energy Profile
A primary function of a catalyst is to lower the activation energy () of a chemical reaction. The activation energy is the minimum energy required for reactants to initiate a chemical reaction.
- In the presence of a catalyst, less energy is needed to start the reaction, meaning more reactant molecules possess the necessary energy to react at a given temperature.
- Crucially, catalysts do not change the overall energy difference () between reactants and products (enthalpy change of the reaction). They only provide an alternative reaction pathway with a lower energy barrier.
- Consequently, catalyzed reactions proceed faster than uncatalyzed ones at the same temperature and reactant concentrations.
The image below illustrates how a catalyst provides an alternative pathway with lower activation energy:
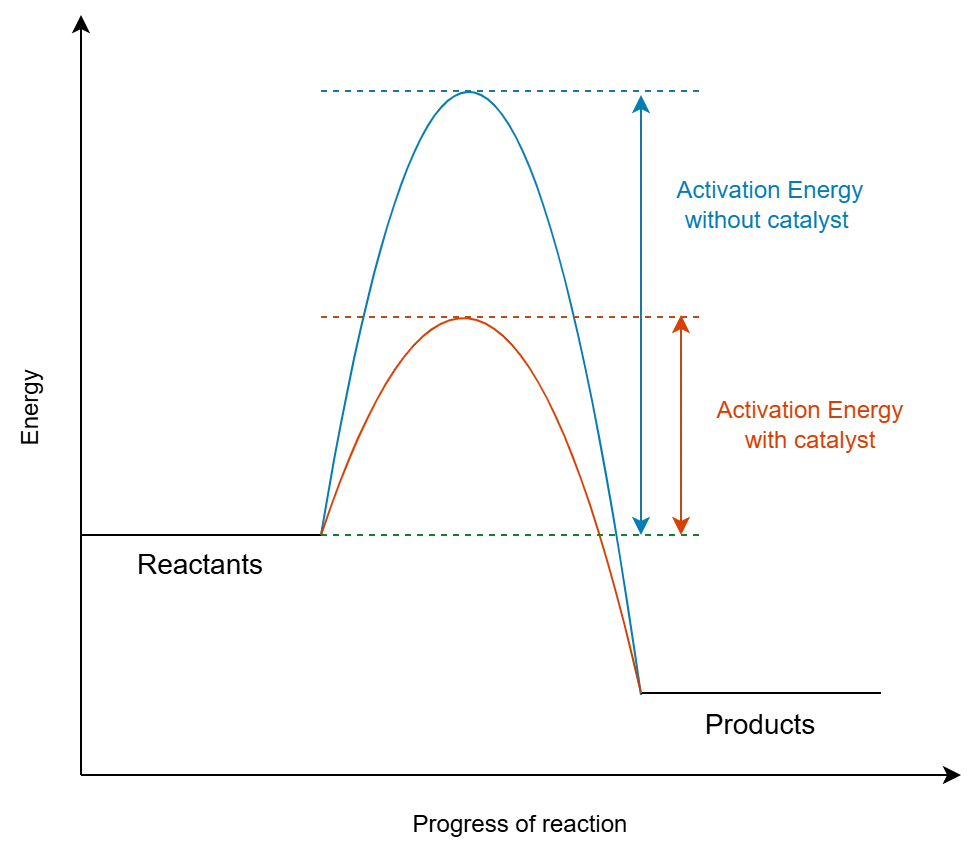
Factors Affecting Catalyst Efficiency
The effectiveness of a catalyst can be influenced by other substances:
- Inhibitors: Substances that decrease the efficiency of a catalyst, making it less effective.
- Promoters: Substances that increase the efficiency of a catalyst. They can also sometimes modify the optimal reaction temperature.
The rate of a catalyzed reaction depends on the frequency with which reactants interact, usually during the slowest step, which often involves the catalyst. While not consumed, catalysts can be deactivated or destroyed by other processes (e.g., poisoning).
20.3.2 Mechanism of Catalysis
Catalysts accelerate reactions by providing an alternative reaction pathway with a lower activation energy. The general steps often involve:
- Adsorption: Reactant molecules bind to the surface of the catalyst.
- Reaction on surface: The catalyst facilitates the breaking and forming of bonds between reactants, often through temporary intermediate species. The specific mechanism depends on the type of catalysis (homogeneous or heterogeneous).
- Desorption: Product molecules detach from the catalyst surface, freeing up the catalyst to participate in further reaction cycles.
Role of Transition Metals in Catalysis
Many transition metals and their compounds are excellent catalysts due to their unique electronic configurations:
- They possess incompletely filled d-orbitals.
- These d-orbitals allow transition metals to readily donate and accept electrons from other molecules. This ability enables them to form temporary bonds with reactants, stabilize intermediate states, and facilitate electron transfer, which is crucial for lowering activation energy.
- Their variable oxidation states also contribute to their catalytic activity.
Catalysts effectively bring reactants together by temporarily bonding with them, which makes it easier and quicker for the reactants to interact and form products.
Example 1: Catalytic Decomposition of Hydrogen Peroxide
This example demonstrates how a heterogeneous catalyst, manganese dioxide, accelerates the decomposition of hydrogen peroxide.
Reaction:
Catalyst: Manganese dioxide ()
Mechanism Steps:
- Adsorption: Hydrogen peroxide () molecules adsorb onto the surface of the solid catalyst.
- Surface Reaction: The surface provides an active site that weakens the O-O bond in , facilitating its breakdown. This involves a lower activation energy pathway compared to the uncatalyzed reaction.
- Intermediate Formation: The decomposition proceeds via the formation of intermediate species involving the surface, though the exact intermediates can be complex (e.g., redox cycles involving manganese).
- Desorption: Water () and oxygen () molecules desorb from the surface as products.
- Catalyst Regeneration: The remains chemically unchanged and is ready to catalyze further decomposition of hydrogen peroxide.
Example 2: Reaction between Peroxodisulphate and Iodide Ions
Transition metals like can act as homogeneous catalysts by changing oxidation states.
Reaction:
Mechanism:
Example 3: Contact Process for Sulfur Trioxide Production
Vanadium(V) oxide is a key catalyst in one of the most important industrial processes for sulfuric acid production.
Reaction:
Catalyst: Vanadium(V) oxide ()
In this process, acts as a heterogeneous catalyst, speeding up the oxidation of sulfur dioxide () to sulfur trioxide (), which is a critical step in the manufacture of sulfuric acid.
Example 4: Autocatalysis
In some reactions, one of the products acts as a catalyst for the reaction itself. This is known as autocatalysis.
Reaction:
The ions produced act as a catalyst, causing the reaction rate to increase as the reaction proceeds.