20.2 Extracting Material from Ores and Alloying
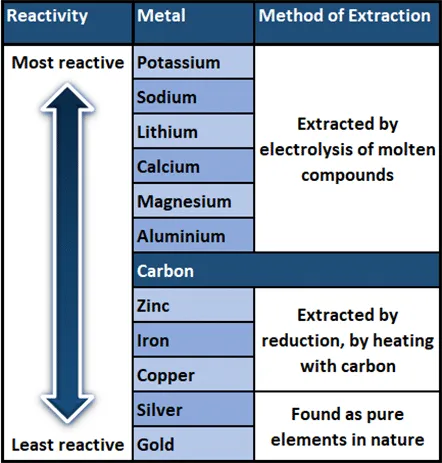
Reactivity and Extraction
The method of extracting a metal is directly determined by its reactivity. More reactive metals require more energy-intensive methods (e.g., electrolysis), while less reactive metals can be extracted through simpler chemical reduction methods.
1. Extraction Process
The initial stages of extracting a metal from its ore involve physical preparation and concentration.
-
Crushing and Grinding: The extracted ore is mechanically broken down into smaller fragments and subsequently reduced to a fine powder to increase its surface area for subsequent chemical processes.
-
Concentration: This step aims to remove unwanted gangue (impurities) from the ore. Commonly employed techniques include:
- Flotation process: Used for sulfide ores, where the ore particles float and gangue sinks.
- Magnetic separation: Used for ores containing magnetic components (e.g., iron ores).
- Gravitational separation: Used for ores where there's a significant density difference between the ore and gangue.
2. Roasting
Roasting is the process where the concentrated ore is heated in the presence of air, typically below its melting point, to convert it into its oxide form. This often releases sulfur dioxide gas.
Examples of Roasting Reactions:
- Zinc blende ():
- Cinnabar (): This is an exception where the metal is directly produced.
- Copper pyrite ore ():
3. Smelting
Smelting is a high-temperature process used to reduce metal ions in the ore to their free metallic state. This is typically achieved by heating the ore with a reducing agent.
- Common Reducing Agents:
- Coke (carbon)
- Carbon monoxide ()
- Hydrogen ()
Examples of Smelting Reactions:
- Iron oxide () reduction:
- Tungsten oxide () reduction:
- Zinc oxide () reduction:
Smelting of Copper Ore (Two Steps)
The smelting of copper ore is a more involved, two-step process:
-
Initial Heating for Matte Formation: The roasted copper ore (containing and ) is heated with coke and sand () at about . The materials melt and separate into two distinct layers:
- Matte: The bottom layer, a molten mixture primarily consisting of and .
- Silicate Slag: The upper layer, formed by the reaction of (from oxidized ) with sand. This slag, being less dense, floats on top and can be removed.
-
Bessemerization: In this crucial step, air is blown through the molten copper matte in a specialized vessel called a Bessemer converter (Figure 20.8).
- Any remaining iron sulfide () is oxidized and removed as slag ().
- Subsequently, cuprous sulfide () is oxidized to form cuprous oxide ().
- The cuprous oxide then reacts with any remaining cuprous sulfide to yield metallic copper.
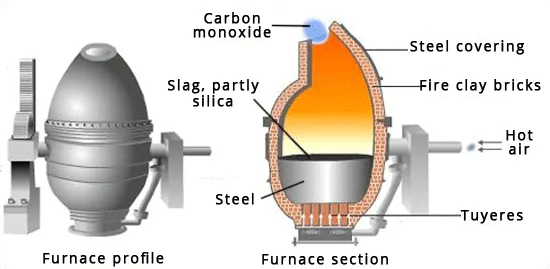
The product from Bessemerization is called blister copper, typically 97-99% pure copper with entrapped bubbles of gas. Bessemerization is also a technique used in steel production from pig iron.
4. Refining or Purification of Metals
The metal obtained from smelting often contains impurities and must be refined to achieve the desired purity.
i) Electrolytic Refining
An electrolytic cell is employed for electrorefining, where:
- The impure metal acts as the anode (positive electrode).
- A sample of pure metal acts as the cathode (negative electrode).
- The electrolyte is a solution containing ions of the metal being refined.
Types Of Electro-Chemical Cells→
Example: Electrolytic Refining of Copper
This process is carried out in an electrolytic tank containing an acidified copper sulfate solution as the electrolyte.
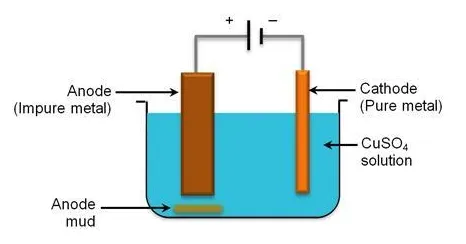
- At the anode (impure copper): Impure copper oxidizes and dissolves, forming ions. More reactive impurities also oxidize and dissolve. Less reactive impurities (like Au, Ag) do not oxidize and fall off as anode mud.
- At the cathode (pure copper): ions from the electrolyte gain electrons and deposit as pure copper atoms, increasing the mass of the pure copper cathode.
ii) Distillation
Metals with relatively low melting points and volatile impurities can be refined by distillation. Examples include arsenic () and mercury (). The metal is heated to its boiling point, vaporized, and then condensed as a purer liquid.
5. Alloying
Alloying is the process of combining a metal with one or more other elements (metals or non-metals) to enhance its properties, such as strength, ductility, corrosion resistance, and hardness.
Methods of Alloying
- Melting and Mixing: The base metal is melted, and the alloying elements are added to the molten metal. The mixture is thoroughly stirred to ensure uniform distribution of the elements. For example, steel is made by adding carbon and other elements like chromium, nickel, and manganese to iron.
- Powder Metallurgy: This involves mixing fine powdered metals and pressing them into a desired shape before heating them (sintering) to bond the particles. This method is useful for creating alloys with specific properties and for metals that are difficult to melt together.
- Mechanical Alloying: This involves repeatedly fracturing and welding a mixture of powder particles in a high-energy mill to produce an alloy. This method is used for creating advanced materials with unique properties, often at the nanoscale.
6. Shaping
After extraction and alloying, metals are shaped into useful forms:
- Casting: The molten metal or alloy is poured into molds to achieve a desired shape, which solidifies upon cooling.
- Forging: The metal or alloy is hammered or pressed while it is hot to reshape it and refine its grain structure, improving its strength.
- Rolling: The metal or alloy is passed through rollers to produce sheets, plates, or other elongated shapes.
- Machining: Cutting, drilling, grinding, and shaping the metal or alloy using various tools to achieve precise dimensions and finishes.
7. Heat Treatment
Heat treatment processes are used to alter the physical and mechanical properties of metals and alloys.
- Annealing: Heating the metal or alloy to a specific temperature and then cooling it slowly (e.g., in a furnace) to remove internal stresses, improve ductility, and make it softer.
- Quenching: Heating the metal or alloy to a high temperature and then cooling it rapidly (e.g., in water or oil) to increase its hardness and strength, but often making it more brittle.
- Tempering: Heating the quenched metal or alloy to a lower temperature than quenching and then cooling it to achieve a desirable balance between hardness, strength, and ductility, reducing brittleness.
Examples of Alloys
- Steel: Iron alloyed with carbon (and often other elements like chromium, nickel, manganese).
- Bronze: Copper alloyed with tin.
- Brass: Copper alloyed with zinc.
Interesting Fact: Purpose of Alloying
A main purpose of making alloys is to decrease the intensity of corrosion of metals.
- For example, bronze (an alloy of 90% copper and 10% tin) is highly resistant to weathering, which is why bronze statues do not get significantly affected by sun and rain.
- Stainless steel (an alloy typically made from 74% iron, 18% chromium, and 8% carbon) does not stain with air or water and exhibits excellent rust resistance due to the formation of a passive chromium oxide layer.
20.2.1 Importance of Alloy Making
- Increased Hardness: Alloys help to significantly increase the hardness of metals compared to their pure forms.
- Reduced Melting Point: The melting point of pure metals is often very high. Alloying can help to reduce the melting point or create a melting point range, which can be advantageous for processing.
- Enhanced Resistance: By alloying, the resistance to corrosion, chemical attack, and adverse weather conditions increases.
- Color Modification: The process of alloying can change the color of metals when two different metals or a metal and a non-metal are mixed.
- Improved Casting Properties: Pure metals tend to solidify and contract significantly when melted, which can lead to defects in casting. Alloys often tend to expand slightly upon solidification or have a narrower solidification range, resulting in better and more uniform castings.