18.9 Carbon-13 NMR Spectroscopy
Carbon-13 Nuclear Magnetic Resonance (C-NMR) is a spectroscopic technique used to determine the structure of organic molecules by identifying the carbon framework. About 1% of all carbon atoms are the isotope, which is NMR active. The most abundant isotope, , is not NMR active.
The principle of C-NMR is based on the magnetic properties of the nucleus. Like a small magnet, a nucleus can align with an external magnetic field () in a low-energy state, or oppose it in a higher-energy state. By supplying energy in the form of radio waves of a specific frequency, the nucleus can be made to "flip" from the more stable to the less stable alignment. This absorption of energy is known as the resonance condition and is detected as a peak in the NMR spectrum.
1. Chemical Shift in C-NMR
The chemical shift () is the position of a signal on the NMR spectrum, measured in parts per million (ppm). It is measured relative to a reference standard, typically Tetramethylsilane (TMS), which is assigned a value of 0 ppm.
- A peak that appears to the left of TMS is described as being downfield.
- The chemical shifts for C-NMR have a much larger range than for proton NMR (H-NMR), typically from 0 to 220 ppm.
- The chemical shift of a carbon atom depends on its electronic environment. Electronegative atoms (like O, N, halogens) or groups attached to or near a carbon atom will "deshield" it, causing its signal to appear further downfield (at a higher ppm value).
2. Deduction of Molecular Structure
Each chemically unique carbon atom in a molecule produces a distinct peak in the C-NMR spectrum. By analyzing the number and position of these peaks, we can deduce key information about the molecule's structure.
- Number of Peaks: The number of signals in the spectrum corresponds to the number of non-equivalent carbon environments.
- Symmetry: Symmetrical molecules will have fewer peaks because multiple carbon atoms are in identical chemical environments. For example, in a molecule with a plane of symmetry, carbons that are mirror images of each other are equivalent and will produce a single peak.
- Chemical Shift Values: The position (ppm value) of a peak indicates the type of carbon atom. Standard chemical shift ranges help identify functional groups.
Table: Approximate Chemical Shift Values for C-NMR
| Type of Carbon Atom | Chemical Shift (, ppm) |
|---|---|
| Alkane (, , ) | 5 - 45 |
| Alkyne () | 65 - 90 |
| C-Halogen () | 10 - 70 |
| C-Oxygen (, alcohol/ether) | 50 - 90 |
| C-Nitrogen () | 40 - 80 |
| Alkene () | 100 - 150 |
| Aromatic () | 110 - 160 |
| Carboxylic Acid/Ester () | 155 - 185 |
| Ketone/Aldehyde () | 190 - 220 |
Note: Combined information from different spectroscopic techniques (like Mass Spectrometry, IR Spectroscopy→, and H-NMR) is often necessary for complete structural elucidation.
3. Steps to Predict the Number of Peaks
- Draw the molecule's structure.
- Look for elements of symmetry, such as planes of symmetry or rotational symmetry.
- Identify equivalent carbons. Carbons that can be interchanged by a symmetry operation are chemically equivalent and will produce only one signal.
- Count the number of unique carbon environments. This count is the predicted number of peaks in the C-NMR spectrum.
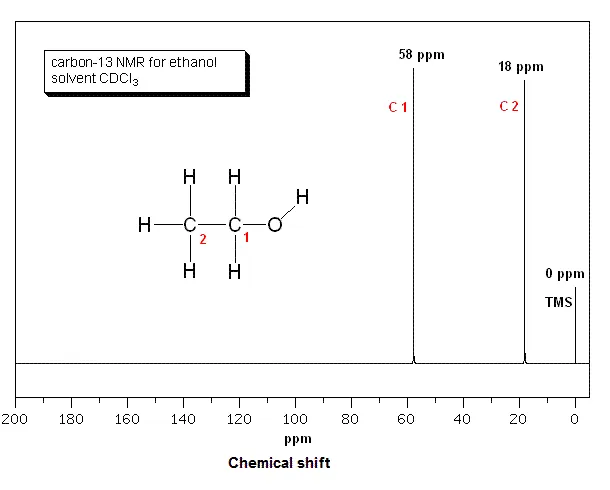
Example 1: Ethanol ()
- Structure:
- Analysis:
- The carbon in the methyl group () is bonded to three hydrogens and the group.
- The carbon in the methylene group () is bonded to two hydrogens, the group, and the highly electronegative oxygen atom.
- These two carbons are in different chemical environments.
- Number of Unique Carbons: 2
- Expected Peaks: 2
- Spectrum Interpretation:
- The carbon is relatively shielded and appears upfield at ~18 ppm.
- The carbon is deshielded by the adjacent oxygen atom and appears downfield at ~58 ppm.
Example 2: Acetone ()
- Structure:
- Analysis:
- The two methyl () groups are chemically equivalent due to the molecule's symmetry. They are both bonded to the same carbonyl carbon.
- The carbonyl () carbon is in a unique environment, double-bonded to an electronegative oxygen atom.
- Number of Unique Carbons: 2
- Expected Peaks: 2
- Spectrum Interpretation:
- The two equivalent carbons appear as a single peak at ~30.8 ppm.
- The carbonyl carbon is highly deshielded and appears far downfield at ~206 ppm.
Example 3: Butane ()
- Structure:
- Analysis:
- The molecule has a plane of symmetry in the middle.
- The two terminal methyl () groups are equivalent.
- The two internal methylene () groups are equivalent.
- Number of Unique Carbons: 2
- Expected Peaks: 2
- Spectrum Interpretation:
- Methyl carbons: Appear around 10-20 ppm.
- Methylene carbons: Appear around 20-40 ppm.
Example 4: Benzene ()
- Structure: A six-membered aromatic ring.
- Analysis:
- Due to the high degree of symmetry in the benzene ring, all six carbon atoms are chemically equivalent.
- Number of Unique Carbons: 1
- Expected Peaks: 1
- Spectrum Interpretation:
- The single peak for the aromatic carbons appears around 120-140 ppm (specifically at 128 ppm).
Q1: How many types of C atoms are present in ethoxyethane (diethyl ether), ? Draw an approximate C-NMR spectrum of this molecule.
A:
- Structure and Symmetry: The molecule is symmetrical around the central oxygen atom.
- Unique Carbons:
- The two methyl () carbons are equivalent.
- The two methylene () carbons, which are attached to the oxygen, are also equivalent.
- Therefore, there are 2 unique types of carbon atoms.
- Approximate Spectrum:
- The spectrum will show 2 peaks.
- The methyl () carbons will appear upfield, around 15 ppm.
- The methylene () carbons are attached to the electronegative oxygen, so they will be deshielded and appear downfield, around 65 ppm.
(Conceptual drawing: A simple spectrum would show two vertical lines on a ppm scale, one at ~15 and a second at ~65.)
Q2: Why are the chemical shifts in C-NMR much greater than in H-NMR?
A: The range of chemical shifts is larger in C-NMR (0-220 ppm) compared to H-NMR (0-12 ppm) primarily because:
- Greater Electronic Variation: Carbon atoms form the backbone of molecules and are directly involved in a wider variety of bonding environments (single, double, triple bonds; bonding to highly electronegative atoms). This creates a much larger range of electronic shielding and deshielding effects compared to protons, which are typically on the periphery of a molecule.
- Polarizability: The electron clouds around carbon are more polarizable than those around hydrogen, leading to larger induced magnetic fields and thus a wider spread of resonance frequencies.