16.2 Types of Errors in Experimental Measurements
Error is the difference between the value obtained in an experiment and the accepted (true) value. In chemical experiments, understanding and minimizing error is crucial for obtaining accurate and reliable results. There are primarily two types of errors.
Random Errors
A random error is the unpredictable, random difference between an observed value and the true value. These errors cause measurements to be scattered around the true value.
Causes of Random Errors:
- Instrument Readability: Difficulty in reading a scale precisely (e.g., estimating the last digit on a burette).
- Environmental Fluctuations: Unpredictable changes in the lab environment, such as temperature fluctuations, air currents, or vibrations.
- Inherent Variability: Natural variations in the quantity being measured.
Characteristics:
- They cause the result to deviate from the accepted value in either direction (either too high or too low).
- The impact of random errors can be reduced by repeating the experiment multiple times and calculating the average of the results.
Systematic Errors
A systematic error is a consistent, repeatable error that is associated with faulty equipment, a flawed experimental design, or human mistakes.
Causes of Systematic Errors:
- Instrumental Errors: An electronic balance that is not zeroed correctly will consistently give readings that are too high.
- Methodological Errors: Not keeping a cap on a spirit burner during a calorimetry experiment leads to alcohol evaporation, resulting in a consistently larger calculated mass loss.
- Personal Errors: Consistently reading a volume from a burette from an angle instead of at eye level (parallax error) will always result in readings that are either too high or too low, depending on the angle.
Characteristics:
- They always shift the result away from the accepted value in the same direction (always too high or always too low).
- Repeating the experiment and averaging the results will not eliminate systematic errors. These errors must be identified and corrected by improving the experimental technique or calibrating equipment.

16.2.1 Propagation of Random Errors
When you perform calculations using data that contains random errors, these errors accumulate or propagate, leading to a larger uncertainty in the final result.
For example, if you measure the length and width of a rectangle to find its area, the uncertainties in your length and width measurements will combine to create an uncertainty in the calculated area.
16.2.2 Systematic Errors and Experimental Design
Careful experimental design is critical to minimize systematic errors. Flaws in the design or procedure can introduce consistent biases that decrease the accuracy and reliability of the results.
Example 1: Spectrophotometry
An experiment is designed to find the concentration of a compound (e.g., Alizarin) using a spectrophotometer. Potential systematic errors include:
- Error in Calibration: If the standard solutions used for calibration have incorrect concentrations, or the calibration curve is plotted inaccurately, all subsequent measurements will be systematically flawed.
- Sample Preparation: Consistently adding slightly more or less sample to the cuvette will lead to systematically incorrect absorbance readings.
- Instrument Performance: The instrument's performance may drift over time due to aging components, leading to consistent errors in measurements.
- Impurity Effect (Matrix Effect): If other substances in the sample absorb light at the same wavelength as the compound of interest, they will interfere with the measurement, causing a systematic error.
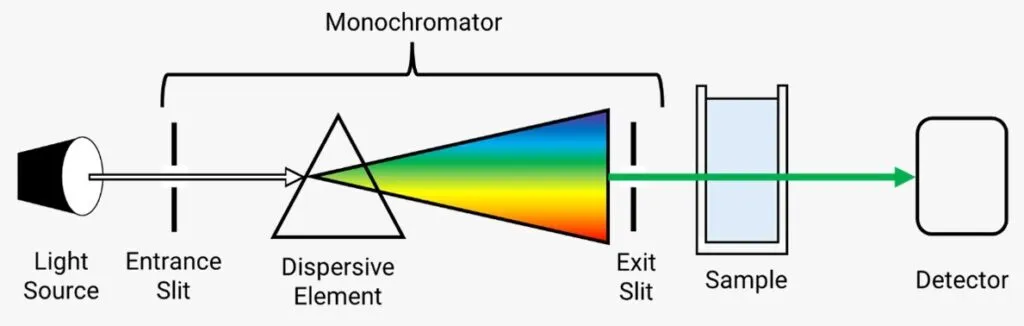
Example 2: Volumetric Titration
In a titration to find the concentration of a reducing agent () with an oxidizing agent (acidified ), the following systematic errors can occur:
- Error in Volume Measurements: Using an uncalibrated burette or pipette, or consistently misreading the meniscus, will introduce a systematic error in all volume readings.
- Impurity in Reagents: If the reagents used (e.g., or ) are impure, the calculated concentration of the analyte will be incorrect.
- Effect of Temperature Fluctuation: Since reaction rates are temperature-dependent, performing titrations at a consistently different temperature than specified can shift the equilibrium and lead to errors in determining the endpoint.
- Incomplete Redox Reaction: If the reaction between the oxidizing and reducing agents does not go to completion (due to inhibitors or inadequate stirring), the endpoint will be inaccurate, leading to an incorrect concentration calculation.
To avoid these errors, it is essential to revise and refine the experimental design and procedure.
Worked Examples
Calculating Propagation of Error
Problem: Calculate the area of a rectangle and its associated uncertainty given the following measurements.
-
Write the given values.
- Length of the rectangle ():
- Uncertainty in length ():
- Width of the rectangle ():
- Uncertainty in width ():
-
Calculate the area.
-
Apply the formula for propagation of uncertainty. Note: A more common formula for multiplication is based on relative uncertainties. The formula provided in the text is a simplified approximation.
-
Show the calculation.
-
State the final result. The final area of the rectangle should be reported with its uncertainty: This means the true area could be as low as or as high as .
Possible Questions/Answers
-
Q: What is the fundamental difference between random and systematic errors? A: Random errors cause measurements to scatter unpredictably around the true value (both higher and lower) and can be minimized by averaging repeated trials. Systematic errors cause measurements to be consistently skewed in one direction (always higher or always lower) and cannot be eliminated by averaging.
-
Q: Why doesn't repeating an experiment and taking the average fix a systematic error? A: Because the error is inherent in the system (e.g., a miscalibrated scale). Every measurement will be off by the same amount and in the same direction, so the average will also be off by that amount. The error must be identified and the experimental method corrected.
-
Q: Give an example of parallax error. A: When reading the volume of a liquid in a burette, if you consistently view the meniscus from above, your reading will always be lower than the true volume. This is a systematic, personal error.