10.3 Acidity of Water, Phenol, and Ethanol
This section explores the acidity of phenols compared to water and ethanol, and details their characteristic reactions including those of related compounds like naphthols.
10.3 Acidity of Water, Phenol, and Ethanol
Acidity refers to the ability of a compound to donate a proton (), which is directly related to the stability of the resulting conjugate base.
Water (): Water is a very weak acid. Its autoionization constant () is , and its acid dissociation constant () is approximately .
Phenol (): Phenol is a stronger acid than water, with a of . This increased acidity is due to the stability of its conjugate base, the phenoxide ion ().
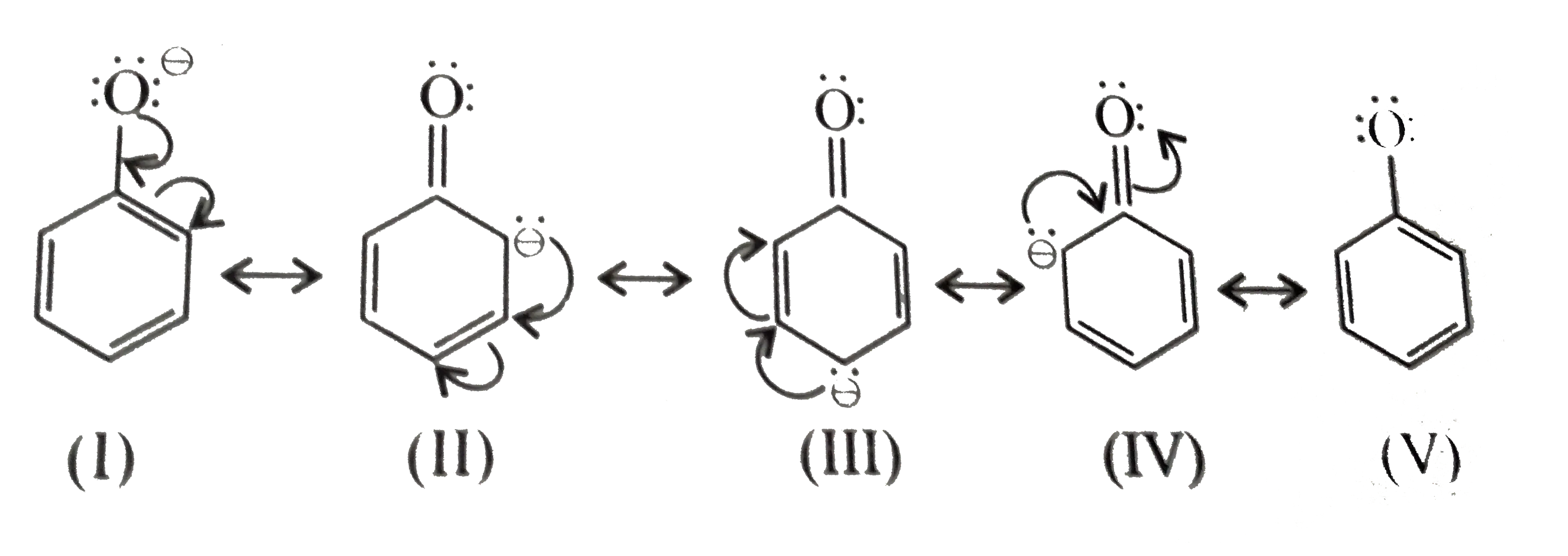
Resonance Stabilization of the Phenoxide Ion
When phenol loses a proton, the resulting negative charge on the oxygen atom is not localized. Instead, it is delocalized (spread out) into the pi-system of the benzene ring through resonance. This distribution of charge stabilizes the phenoxide ion, making the formation of the ion (and thus, the donation of the proton) more favorable compared to water or ethanol.
Dissociation of Phenol:
Resonance Structures of Phenoxide Ion:
| Compound | Formula | Value | Relative Acidity | Reason for Stability of Conjugate Base |
|---|---|---|---|---|
| Phenol | Weakly Acidic | Resonance stabilization of phenoxide ion | ||
| Water | Very Weak Acid | No resonance stabilization for | ||
| Ethanol | Least Acidic | Electron donating ethyl group destabilizes ethoxide |
Reactions of Phenol
Phenol undergoes several characteristic reactions due to the activating effect of the hydroxyl group.
Reaction with Sodium Hydroxide
Phenol reacts with sodium hydroxide to form sodium phenoxide (sodium phenolate), a salt.
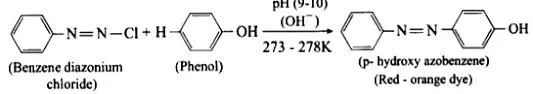
Reaction with Diazonium Salt (Azo Coupling)
This reaction forms azo-dyes, which are colored synthetic compounds containing an azo group () that links two aromatic rings.
Procedure:
- Phenol is dissolved in a sodium hydroxide solution, forming the highly reactive sodium phenoxide ion.
- The solution is cooled in ice to about 0-5 °C.
- A cold solution of benzene diazonium chloride is added slowly.
Observation: A yellow-orange solution or precipitate is formed.
Product: The product is an azo compound, such as p-hydroxyazobenzene.
Overall Reaction:
Mechanism:
10.5 Reactions of Naphthols
Naphthols are derivatives of naphthalene containing a hydroxyl group. They behave chemically like simple phenols.
1. Reaction with Sodium Hydroxide
Like phenols, naphthols are acidic and react with strong bases to form sodium naphthoxide salts.
1-Naphthol (α-naphthol):
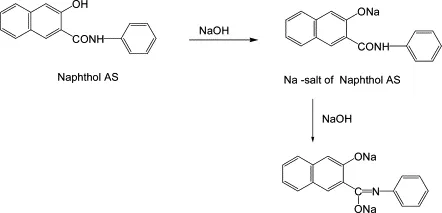
2-Naphthol (β-naphthol):
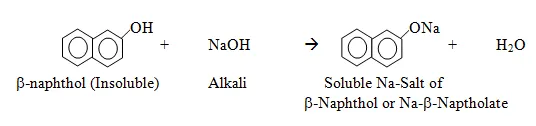
2. Reaction with Bromine Water (Bromination)
Naphthols undergo electrophilic substitution and decolorize bromine water. The position of substitution depends on the isomer.
1-Naphthol: Substitution occurs at the activated 2- and 4-positions.

2-Naphthol: Substitution occurs at position 1, which is adjacent to the -OH group. Position 3 is less reactive, and other positions are sterically hindered by the fused ring.

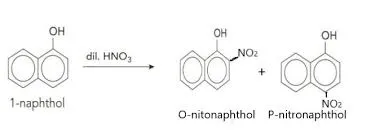
3. Reaction with Nitric Acid (Nitration)
Nitration occurs similarly to bromination.
1-Naphthol: Reaction with nitric acid yields a mixture of 2-nitro-1-naphthol and 4-nitro-1-naphthol.
4. Reaction with Diazonium Chloride (Azo Coupling)
Naphthols react with diazonium salts to give intensely colored azo compounds. The reaction is performed under the same cold, basic conditions as with phenol.
1-Naphthol: The diazonium salt attacks at position 4.
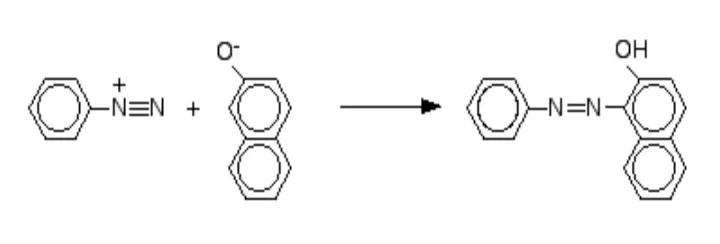
2-Naphthol: The diazonium salt attacks at position 1.
