10.2 Preparation of Phenol
This section describes the laboratory and industrial methods for synthesizing phenol. While the laboratory method involves a two-step process from an aromatic primary amine (aniline), several industrial methods are used to produce phenol on a large scale.
Preparation from Phenylamine (Aniline)
The preparation of phenol from phenylamine involves two main stages: diazotization and hydrolysis.
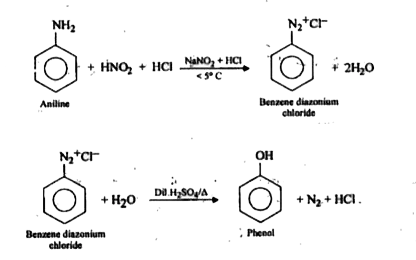
Step 1: Formation of Benzene Diazonium Salt (Diazotization)
Phenylamine (aniline) is first converted into a diazonium salt.
Reactants:
- Phenylamine ()
- Nitrous acid (), generated in situ from sodium nitrite () and hydrochloric acid ()
Conditions: The reaction must be carried out at a temperature below (usually ) to prevent decomposition of the unstable diazonium salt.
The amine group () is converted into a diazonium group (). The reaction is:
Step 2: Hydrolysis of the Diazonium Salt
The benzene diazonium salt is converted to phenol by gentle warming with water.
Conditions: The aqueous solution is warmed to above .
Reaction: The diazonium group is replaced by a hydroxyl group () from water. Nitrogen gas is evolved.
Overall Reaction Scheme
The complete two-step synthesis from aniline to phenol is shown below.
Industrial Preparation of Phenol
In the FBISE curriculum, several industrial methods are essential for understanding the large-scale production of phenol.
1. From Chlorobenzene (Dow's Process)
Chlorobenzene is heated with at and atm pressure to form sodium phenoxide, which is then treated with to release phenol.
2. From Sodium Benzene Sulfonate
Sodium benzene sulfonate is fused with solid at to yield sodium phenoxide.
3. From Cumene (Isopropylbenzene)
This is a modern commercial method. Cumene is oxidized by air to cumene hydroperoxide, which is then decomposed by dilute into phenol and acetone.
Summary
Phenol is synthesized from phenylamine (aniline) in two steps:
- Diazotization: Phenylamine reacts with (from ) below to form benzene diazonium chloride.
- Hydrolysis: Benzene diazonium chloride is warmed above to form phenol, nitrogen gas, and HCl.
Commercially, phenol is prepared via Dow's Process (from chlorobenzene), sulfonation (from benzene sulfonic acid), or the Cumene process.
The diazonium salt serves as a key intermediate in organic synthesis. For further details on the reactivity of phenol, see .