8.3 Reactivity of Benzene: Substitution, Addition, and Oxidation Reactions
Benzene is a unique organic compound characterized by its highly stable delocalized pi () electron ring. This aromatic stability influences its reactivity, leading to distinct types of reactions compared to typical unsaturated hydrocarbons like alkenes. Benzene primarily undergoes electrophilic substitution reactions, but can also participate in addition and oxidation reactions under specific, often vigorous, conditions.
8.3.1 Electrophilic Aromatic Substitution (EAS) Reactions
Electrophilic aromatic substitution reactions are the most characteristic reactions of benzene. They involve an electrophile attacking the high electron density of the delocalized -electron ring, resulting in the replacement of a hydrogen atom on the benzene ring by the electrophile. The delocalized -electron system is restored in the final product, preserving the aromatic stability.
General Mechanism of Electrophilic Aromatic Substitution Reactions:
EAS reactions involve three main steps:
-
Generation of the Electrophile: An electrophile () is generated from a suitable reagent, often with the help of a Lewis acid catalyst (e.g., or ).
-
Attack of Electrophile on Benzene (Formation of Arenium Ion): The -electrons of the benzene ring attack the electrophile, forming a positively charged intermediate known as an arenium ion (also called a sigma () complex). In this step, the aromaticity of the benzene ring is temporarily broken.
-
Removal of Hydrogen (Deprotonation): A Lewis base (often the conjugate base of the catalyst or the catalyst itself) removes a hydrogen ion () from the arenium ion. This step restores the delocalized -electron system, regenerating the aromatic ring and forming the substituted benzene product. The catalyst is also regenerated.
Key Information: The delocalized -electron system in benzene is far more stable than the localized -bonds in alkenes. This is why benzene always requires a catalyst for electrophilic substitution reactions, unlike alkenes which readily undergo addition reactions.
Specific Electrophilic Aromatic Substitution Reactions
Halogenation
Halogenation is the reaction where a hydrogen atom on the benzene ring is replaced by a halogen atom (e.g., , ). This reaction typically requires a Lewis acid catalyst, such as iron(III) halide () or aluminum halide ().
General Reaction:
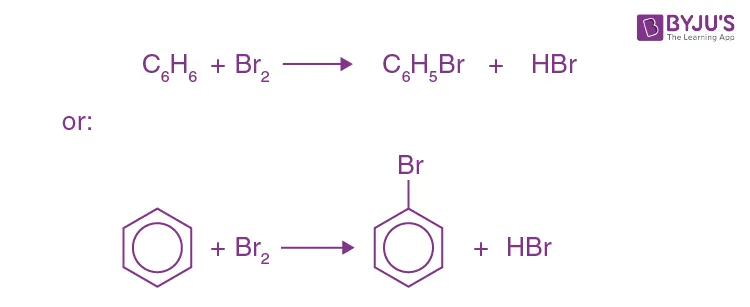
Example: Bromination of Benzene When benzene reacts with bromine in the presence of iron(III) bromide (), bromobenzene is formed.
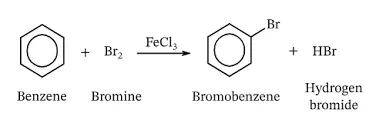
Mechanism of Bromination:
-
Generation of Electrophile (): Bromine () acts as a Lewis base, donating electrons to the Lewis acid , generating the highly reactive bromonium ion ().
-
Formation of Arenium Ion (-Complex): The -electrons of the benzene ring attack the bromonium ion (), forming a carbocation intermediate (arenium ion), which has lost its aromaticity temporarily.
-
Deprotonation/Regeneration of Catalyst: The ion (a Lewis base) abstracts a proton () from the arenium ion, restoring the aromatic ring and regenerating the catalyst.
Chlorination of Benzene: Similar to bromination, chlorination occurs with chlorine gas in the presence of a catalyst like , producing chlorobenzene and hydrogen chloride.
Mechanism follows the same three steps as bromination, substituting with and with .
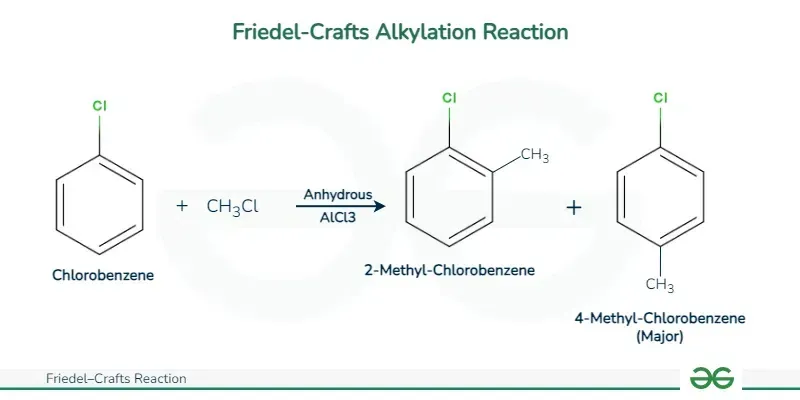
Halogenation of Alkylarenes (e.g., Methylbenzene): Alkyl substituents (like methyl groups) are ring activating and ortho/para directing. They increase electron density at positions 2 (ortho) and 4 (para) on the benzene ring, making these positions more susceptible to electrophilic attack. Hydroxyl () and amino () groups similarly activate the ring.
Example: Chlorination of methylbenzene (toluene) produces a mixture of 2-chlorotoluene and 4-chlorotoluene.
In excess chlorine, multiple substitutions can occur, leading to products like 2,4-dichlorotoluene, 2,6-dichlorotoluene, and 2,4,6-trichlorotoluene.
Side Chain Halogenation: In contrast to ring halogenation, if chlorine reacts with an alkyl side chain of an alkylbenzene in the presence of ultraviolet (UV) light (or strong sunlight), a free-radical substitution mechanism occurs on the side chain, not the ring.
With excess chlorine, all hydrogen atoms on the methyl side chain can be replaced by chlorine atoms.
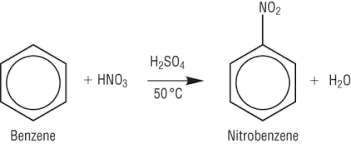
Nitration
Nitration introduces a nitro group () to the benzene ring. The electrophile for this reaction is the nitronium ion (), generated from a mixture of concentrated nitric acid () and concentrated sulfuric acid (), known as the 'nitrating mixture'. The reaction is typically carried out by refluxing benzene at .
General Reaction:
Mechanism of Nitration:
- Generation of Nitronium Ion (): Sulfuric acid protonates nitric acid, which then dehydrates to form the nitronium ion. Or more generally:
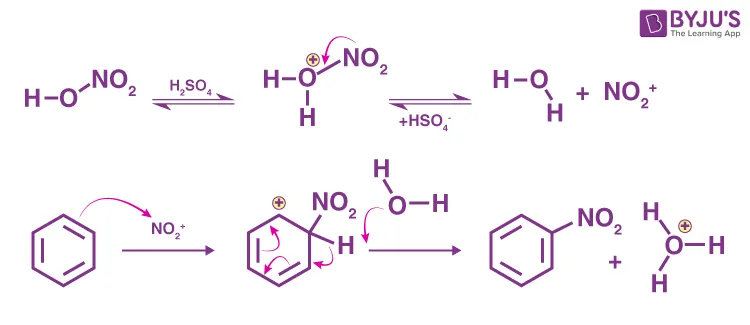
-
Formation of Arenium Ion (-Complex): The benzene -electrons attack the nitronium ion, forming the arenium ion intermediate. This intermediate is resonance stabilized.
-
Deprotonation/Regeneration of Catalyst: A base (typically ) removes a proton from the arenium ion, restoring the aromaticity and forming nitrobenzene. Sulfuric acid is regenerated.
Friedel-Crafts Reactions
Discovered by Charles Friedel and James Crafts in 1877, these reactions introduce alkyl or acyl groups to the benzene ring, catalyzed by Lewis acids like aluminum chloride ().
i. Alkylation (Friedel-Crafts Alkylation)
Introduction of an alkyl group (e.g., methyl, ethyl) into the benzene ring by reacting benzene with a chloroalkane (alkyl halide) in the presence of a Lewis acid catalyst like .
General Reaction:
Example: Reaction of benzene with chloromethane to form methylbenzene (toluene).
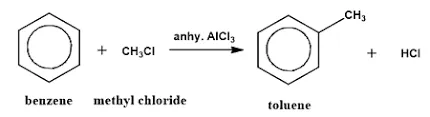
Mechanism of Friedel-Crafts Alkylation:
-
Generation of Electrophile (): The alkyl halide reacts with the Lewis acid catalyst () to generate a carbocation (alkyl cation), which acts as the electrophile. Example: Generation of methyl cation ().
-
Formation of Arenium Ion: The benzene -electrons attack the alkyl cation, forming the arenium ion. This ion is resonance stabilized.
-
Deprotonation/Regeneration of Catalyst: The ion removes a proton from the arenium ion, restoring aromaticity and regenerating .
ii. Acylation (Friedel-Crafts Acylation)
Introduction of an acyl group () into the benzene ring by reacting benzene with an acyl halide () in the presence of a Lewis acid catalyst (). The product is a phenyl ketone.
Mechanism of Friedel-Crafts Acylation:
-
Generation of Electrophile (Acylium Ion): The acyl halide reacts with the Lewis acid catalyst () to generate a resonance-stabilized acylium ion (), which is the electrophile.
-
Formation of Arenium Ion: The benzene -electrons attack the acylium ion, forming the arenium ion intermediate.
-
Deprotonation/Regeneration of Catalyst: A base () removes a proton from the arenium ion, restoring aromaticity and forming the phenyl ketone. is regenerated.
Key Information: Aluminum chloride () is often referred to as a halogen carrier in Friedel-Crafts reactions because it helps generate the electrophilic halogen species.
8.3.2 Addition Reactions
Benzene, despite its apparent unsaturation, is generally reluctant to undergo addition reactions under normal conditions. This is due to the inherent stability of its delocalized -electron aromatic system. Breaking this stable system requires harsh conditions (high temperature, high pressure, strong reagents).
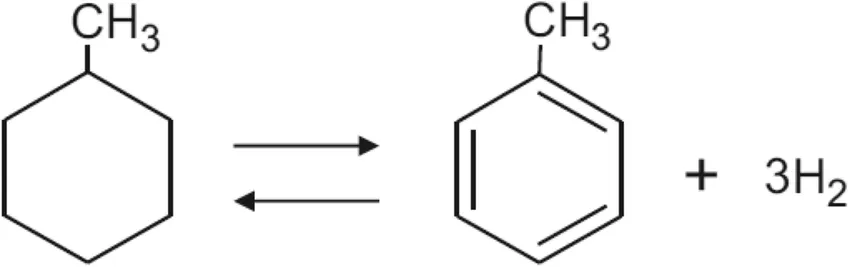
Hydrogenation
Hydrogenation is the addition of hydrogen molecules across the carbon atoms of the benzene ring, converting it into cyclohexane. This reaction breaks the aromaticity and requires specific conditions:
- Catalysts: Nickel (\mathrm{Ni), Platinum (), or Palladium ()
- Temperature:
- Pressure:
General Reaction:
Role of Catalyst: The catalyst provides a surface area that breaks hydrogen molecules into individual hydrogen atoms. It also interacts with the benzene molecule, weakening its -electron system and making it more reactive. Six hydrogen atoms then add to the six carbon atoms of the benzene molecule, forming cyclohexane and releasing it from the catalyst surface.
Example: Hydrogenation of methylbenzene yields methylcyclohexane under similar conditions.
Difference Between Addition Reactions and Electrophilic Substitution Reactions
The fundamental difference lies in the fate of the intermediate carbocation formed after the initial electrophilic attack:
| Feature | Electrophilic Aromatic Substitution | Addition Reactions (e.g., Alkenes) |
|---|---|---|
| Initial Attack | Electrophile attacks -system | Electrophile attacks -system |
| Intermediate | Arenium ion (retains some unsaturation) | Bridged halonium ion or carbocation (localized positive charge) |
| Second Step | Loss of to restore aromaticity | Attack by nucleophile to form a saturated product |
| Aromaticity | Restored in the final product | Destroyed (if aromatic) or leads to saturated product |
| Product | Substituted aromatic compound | Adduct (saturated compound) |
| Conditions for Benzene | Requires catalyst, relatively mild | Requires harsh conditions (high T/P, strong reagents) |
8.3.3 Oxidation Reactions
Benzene and other alkanes are generally resistant to oxidation by common oxidizing agents. However, they can be oxidized under certain conditions. In alkylbenzenes, the aliphatic side chain is often more susceptible to oxidation than the aromatic ring itself, especially under prolonged heating or strong oxidizing conditions.
The presence of the benzene ring activates the alkyl side chain towards oxidation. For instance, while alkanes are not easily oxidized by potassium manganate(VII), the methyl side-chain of methylbenzene is readily oxidized by hot alkaline or hot acidic potassium manganate(VII) () to produce benzoic acid.
The oxidation typically targets the carbon atom of the alkyl side-chain closest to the benzene ring (the benzylic carbon), converting it into a carboxylic acid group (). Longer alkyl chains will have the benzylic carbon oxidized to a carboxylic acid, and other carbons may be oxidized to carbon dioxide.
Example: Oxidation of Methylbenzene to Benzoic Acid
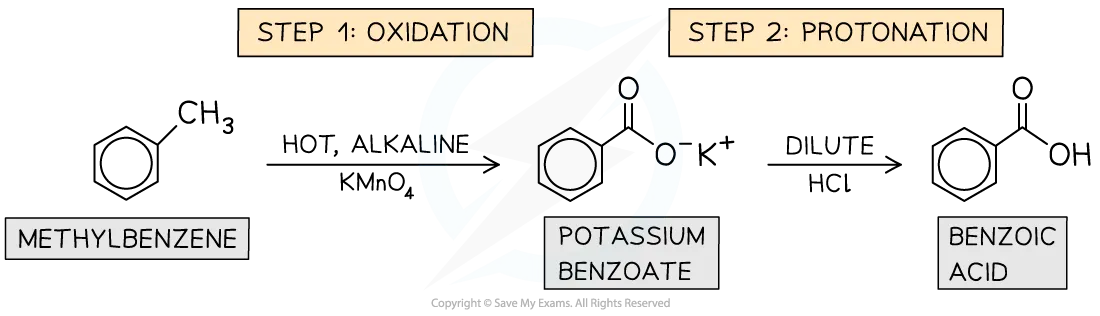
For alkyl chains longer than methyl, all carbon atoms in the side chain, except the benzylic carbon (which forms the carboxylic acid group), are typically oxidized to carbon dioxide ().
Mechanism of Methylbenzene Oxidation to Benzoic Acid:
The mechanism involves a series of free-radical and carbocation intermediates:
-
Hydrogen Abstraction & Benzyl Cation Formation: Hot alkaline abstracts a benzylic hydrogen atom as a free radical, forming a benzyl radical. This radical then reacts with an odd-electron intermediate of to form a benzyl cation.
-
Hydroxyl Group Addition: The benzyl cation abstracts a hydroxyl group () from potassium hydrogenmanganate, forming an unstable alcohol.
-
Further Oxidation: The alkaline continues to oxidize the benzylic carbon, first to a diol, then to benzaldehyde, and finally to benzoic acid.