8.2 The Structure of Benzene
Initial Challenges in Determining Benzene's Structure
After its discovery in 1835, the structure of benzene () remained a puzzle for chemists for forty years. Elemental analysis revealed an unusually low hydrogen-to-carbon ratio (H:C = 1:1), indicating a high degree of unsaturation. Many chemists initially proposed open-chain structures containing carbon-carbon double and triple covalent bonds.
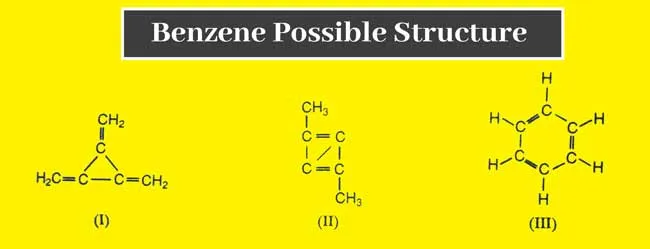
However, these structures were inconsistent with the observed high stability and unique chemical properties of benzene.
Kekulé's Structure of Benzene
In 1865, the German chemist August Kekulé proposed the first ring (cyclic) structure for benzene. This idea came to him in a dream, where he envisioned atoms forming chains that then transformed into rings, resembling a snake biting its tail.
- Key Features of Kekulé's Model:
- Benzene is a cyclic molecule with six carbon atoms arranged in a ring.
- Each carbon atom is bonded to one hydrogen atom.
- The ring contains three alternating carbon-carbon double bonds.
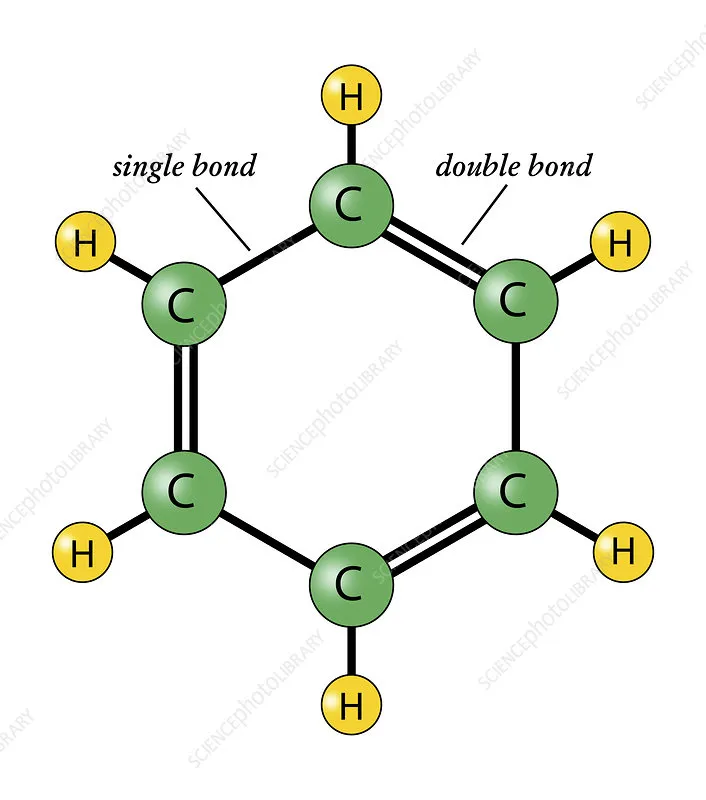
Kekulé further proposed that the double bonds in benzene rapidly alternate their positions, making the two possible structures indistinguishable. This rapid alternation suggested that the pi () electrons are free to move over all six carbon atoms.
Limitations of Kekulé's Structure
Despite its evolutionary nature, Kekulé's model faced several challenges due to its inability to fully explain benzene's observed properties:
- Reactivity with Alkenes: If benzene contains double bonds like alkenes, why does it not react in the same way (e.g., undergoing addition reactions readily)?
- Bromine Addition: Why cannot benzene add three moles of bromine, as would be expected for a molecule with three distinct double bonds?
- Reaction Preference: Why does benzene favor substitution reactions over addition reactions?
These inconsistencies led chemists to seek more accurate models for the structure of benzene.
Resonance Model of Benzene
The concept of resonance provides a more accurate description of benzene's structure, addressing the limitations of Kekulé's model. Linus Pauling, in the 1930s, significantly developed the theory of resonance and applied it to organic molecules.
- Definition of Resonance: Resonance is the spreading or delocalization of electron pairs (either lone pairs or pi electron pairs) across several adjacent atoms within a molecule or ion.
- Resonance Hybrid: When a single Lewis structure cannot accurately represent the true distribution of electrons, the molecule is said to exhibit resonance. Its actual structure is a resonance hybrid - an average of all possible contributing Lewis structures.
- Canonical Structures: The different possible Lewis structures that contribute to the resonance hybrid are called resonance contributing structures or canonical structures. These are hypothetical and do not exist independently; only the resonance hybrid is real.
For benzene, the true structure is an average of the two Kekulé structures, often represented by a hexagon with an inscribed circle. The circle symbolizes the six delocalized pi () electrons shared over all six carbon atoms.
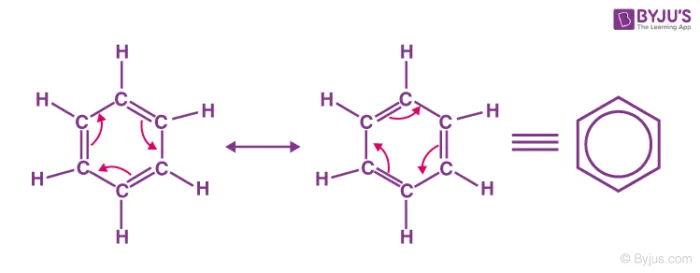
- Why Kekulé structures are still used:
- They justify the tetravalent nature of carbon and the electron count.
- They easily represent the movement of electrons in reaction mechanisms.
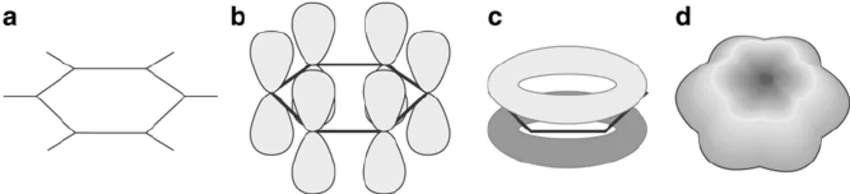
Atomic/Molecular Orbital Model of Benzene
The atomic/molecular orbital model, also developed with concepts of hybridization and resonance by Linus Pauling, provides the most comprehensive explanation for benzene's structure.
- Carbon Hybridization: Each of the six carbon atoms in the benzene ring exhibits hybridization.
- Sigma () Bonds:
- Each carbon forms two bonds with adjacent carbon atoms via orbital overlap.
- Each carbon forms one bond with a hydrogen atom via orbital overlap.
- Molecular Geometry: The carbon skeleton forms a regular hexagon, with all C-C-C and H-C-C bond angles being , indicating a planar structure.
- Bond Lengths:
- Experimental measurements show that the carbon-carbon bond length in benzene is .
- This value is intermediate between a typical carbon-carbon single bond () and a carbon-carbon double bond (). This confirms that the C-C bonds in benzene are neither purely single nor purely double, but possess partial double bond character.
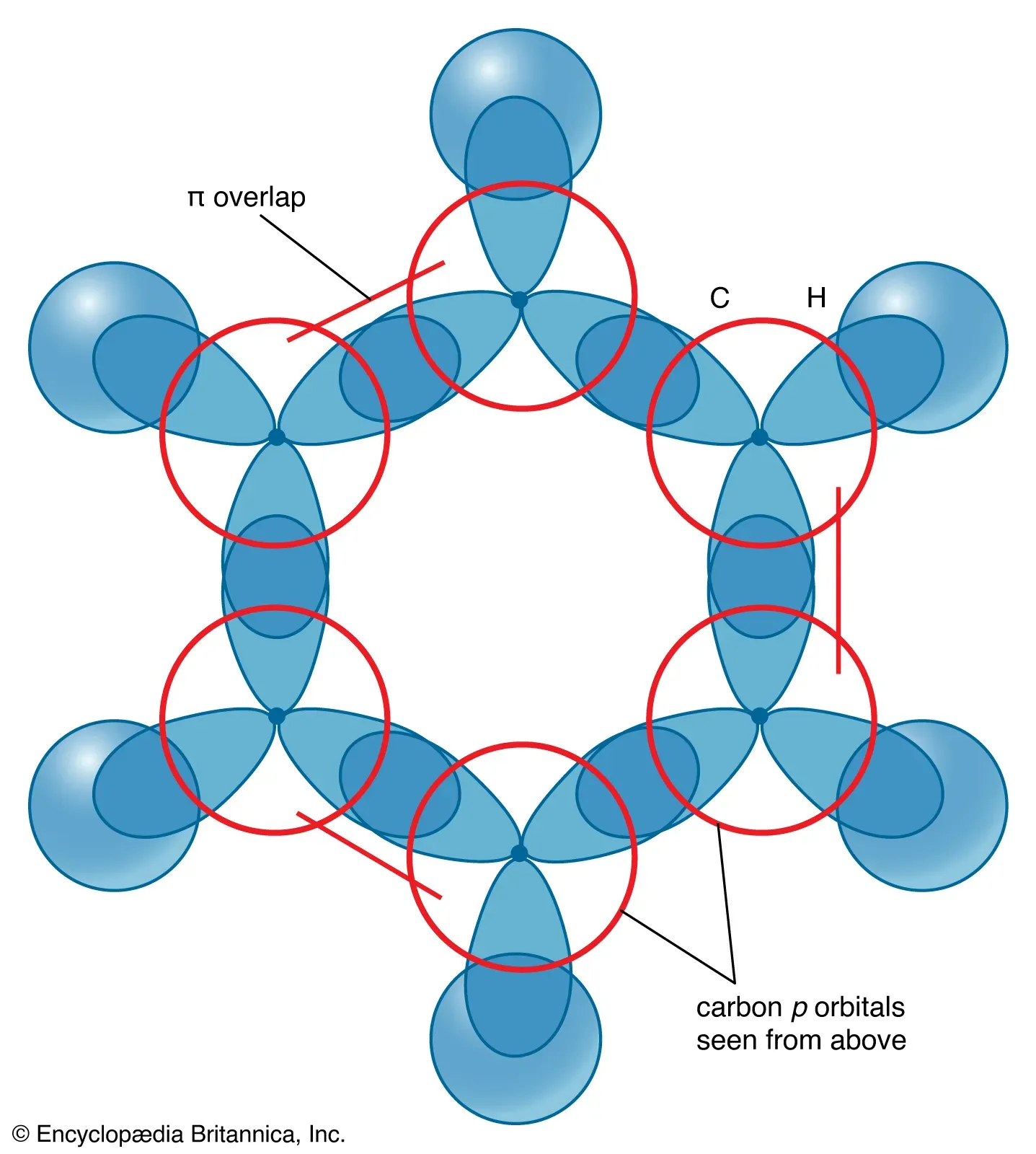
- Pi () Electron System:
- Each -hybridized carbon atom also possesses one unhybridized orbital.
- These six orbitals are perpendicular to the plane of the carbon ring and each contains one electron.
- These six orbitals overlap laterally and continuously around the entire ring, forming a delocalized electron cloud.
- This electron system creates two torus-shaped (doughnut-shaped) regions of electron density: one located above the plane of the ring and another below it.
Resonance Energy and Exceptional Stability of Benzene
The delocalization of -electrons in benzene contributes to its unexpectedly high stability, quantified as resonance energy (or delocalization energy).
- Definition: Resonance energy is the difference in energy between the actual benzene molecule and a hypothetical structure with localized double bonds (like 1,3,5-cyclohexatriene).
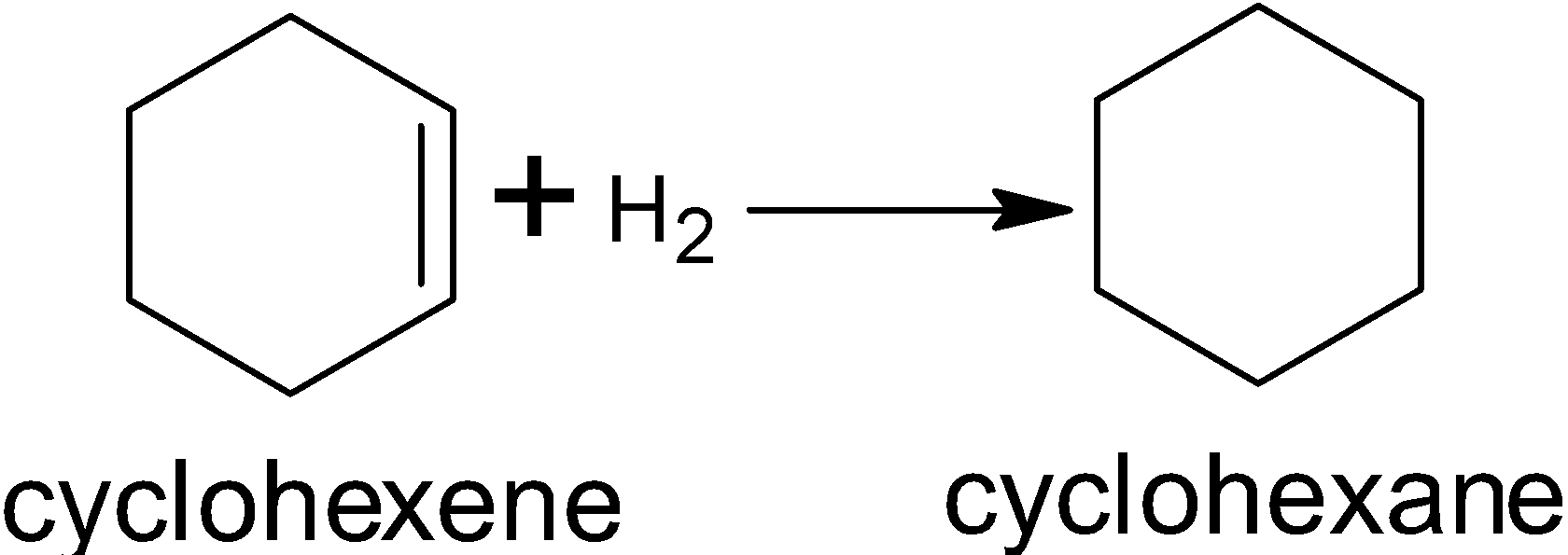
- Measurement: Resonance energy is typically determined by comparing the experimental enthalpy of hydrogenation of benzene with the theoretical enthalpy calculated for a hypothetical cyclohexatriene.
Worked Examples
Calculation of Benzene's Resonance Energy
To calculate the resonance energy of benzene, we compare the enthalpies of hydrogenation:
-
Hydrogenation of Cyclohexene:
- The experimental enthalpy of hydrogenation for cyclohexene (containing one double bond) is:
-
Theoretical Hydrogenation of Hypothetical 1,3,5-Cyclohexatriene:
- If benzene were a simple 1,3,5-cyclohexatriene with three isolated double bonds (like Kekulé's initial idea), its enthalpy of hydrogenation would theoretically be three times that of cyclohexene:
-
Experimental Hydrogenation of Benzene:
- The experimentally observed enthalpy of hydrogenation for benzene to cyclohexane is significantly less exothermic:
-
Calculation of Resonance Energy:
- The difference between the theoretical and experimental values represents the resonance energy, which is the extra stability gained from electron delocalization:
- Often quoted as a positive value, indicating the amount of energy released upon hydrogenation is less than expected, meaning benzene is more stable by that amount. So, the extra stability (resonance energy) is .
This significant difference of highlights the enhanced stability of benzene due to the delocalized -electron system.
Possible Questions and Answers
Q: Draw the two Kekulé structures of benzene. How did Kekulé switch to a closed structure of the benzene molecule, although other chemists tried open structures for it?
A: The two Kekulé structures show alternating single and double bonds within a six-membered carbon ring.
Kekulé's breakthrough to a cyclic structure was reportedly inspired by a dream of a snake biting its tail, symbolizing a ring. This imaginative leap allowed him to move beyond the limitations of proposed open-chain structures, which could not explain benzene's observed stability and unusual chemical inertness toward addition reactions characteristic of alkenes.
Q: Define resonance in benzene. Why is benzene exceptionally stable than alkenes?
A: Resonance in benzene refers to the delocalization of its six -electrons over all six carbon atoms in the ring. Instead of fixed alternating single and double bonds, the actual structure is a resonance hybrid of two contributing Kekulé structures.
Benzene is exceptionally stable compared to alkenes (like a hypothetical 1,3,5-cyclohexatriene) due to this -electron delocalization. This enhanced stability is quantified as resonance energy. The delocalized electron cloud provides a lower energy state than if the electrons were localized in discrete double bonds, making the molecule less reactive and more stable.